The Critical Role of SPE in Pharmacokinetic Studies
Solid-phase extraction (SPE) has become an indispensable tool in modern pharmacokinetic research, particularly for drug metabolite analysis. As noted in pharmaceutical research literature, SPE is “widely used for the preparation of biological samples for further analysis in areas as diverse as clinical chemistry, forensic science, and biomedical and pharmaceutical research.” This popularity stems from its ability to achieve high selectivities and recoveries while minimizing hazardous solvent consumption.
In pharmacokinetic studies during drug development, modern drug candidates are often very potent substances, requiring relatively low doses in preclinical and clinical studies. As a result, assay sensitivity becomes a major goal—sensitivity must be high enough to allow estimation of the terminal plasma half-life in vivo. To obtain such sensitivities, clean plasma extracts are essential, making SPE a critical component of the analytical workflow.
The versatility of SPE is particularly valuable for metabolite analysis, where researchers must isolate and quantify not only parent drugs but also their various metabolic products. As documented in forensic applications, SPE procedures have been successfully applied to isolate a wide variety of drugs and metabolites, including acidic, neutral, and basic compounds from complex biological matrices.
Biological Matrix Challenges in Metabolite Analysis
Complex Matrix Composition
Biological samples present formidable challenges for analytical chemists. Plasma, urine, and tissue samples contain numerous endogenous compounds that can interfere with metabolite detection and quantification. These matrices include proteins, lipids, salts, and various metabolic byproducts that can co-elute with target analytes or cause ion suppression in mass spectrometry.
As noted in SPE methodology literature, extraction of biological samples before injection into analytical systems serves multiple objectives: concentration, clean-up, prevention of analytical column clogging, and elimination of protein binding. The complexity of biological matrices requires careful consideration of matrix composition when developing SPE methods, as identifying and eliminating interferences requires good knowledge of the matrix components.
Protein Binding and Recovery Issues
Many drugs and their metabolites exhibit significant protein binding in biological matrices, particularly in plasma and serum. This binding can reduce extraction efficiency and complicate quantitative analysis. SPE methods must be designed to disrupt these interactions effectively while maintaining analyte integrity.
Research has shown that SPE procedures can be developed to handle highly protein-bound compounds. For instance, studies have demonstrated successful extraction of the highly protein-bound dopamine uptake inhibitor GBR 12909 from biological matrices using optimized SPE protocols.
SPE Cleanup Strategies for Metabolite Analysis
Method Development Strategy
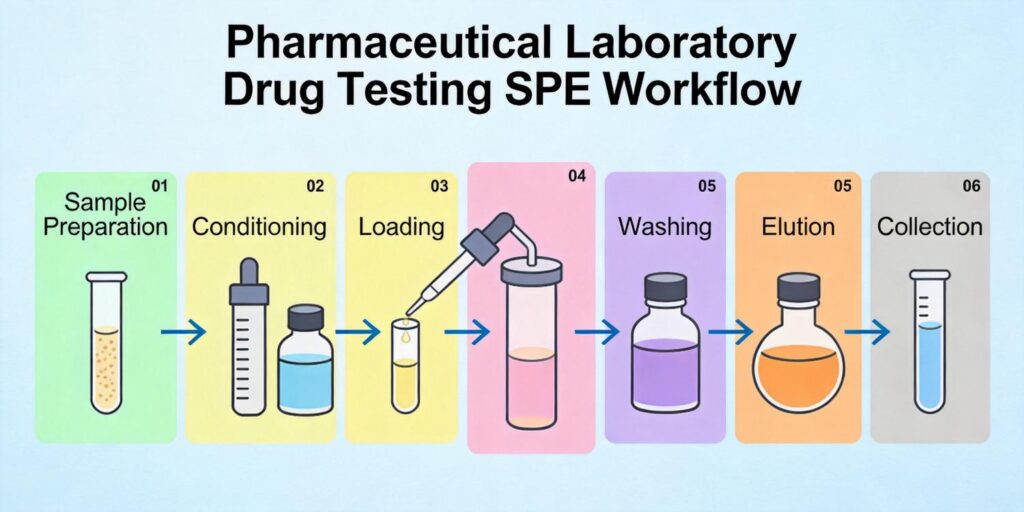
Developing effective SPE methods for metabolite analysis requires a systematic approach. The fundamental steps for SPE in adsorption mode include: prewash (if necessary), preconditioning, sample loading, washing, and elution. Each step must be optimized based on the chemical properties of both the parent drug and its metabolites.
A rational method development strategy should include:
- Analyte Characterization: Understanding the structure, pKa, polarity, functional groups, and stability of both parent drugs and metabolites
- Matrix Characterization: Identifying potential interferences and understanding matrix pH, ionic strength, and variability
- Sorbent Selection: Choosing appropriate SPE phases based on analyte properties
- pH Optimization: Adjusting sample pH to control ionization states and retention characteristics
- Solvent Optimization: Selecting appropriate wash and elution solvents
Mixed-Mode SPE Approaches
For comprehensive metabolite analysis, mixed-mode SPE cartridges that provide both hydrophobic and ion-exchange interactions have proven particularly effective. These sorbents allow for selective extraction based on multiple retention mechanisms, enabling the isolation of metabolites with diverse chemical properties from a single sample.
As documented in toxicological analysis, “the strategy of a mixed-mode cartridge providing hydrophobic and cation exchange interactions, combined with a pH-dependent sample application and extraction, can give high recoveries of analytes from plasma, urine, whole blood, and tissues.” The resulting SPE eluates are easily amenable to subsequent GC and HPLC analysis, with chromatograms showing minimal interference from endogenous matrix components.
Automation and High-Throughput Considerations
Modern pharmacokinetic studies often require analysis of large sample sets, making automation increasingly important. SPE lends itself well to automation, which can increase throughput and substantially reduce manual labor. Automated SPE systems in 96-well plate formats have become standard in many laboratories, particularly when coupled with LC-MS/MS analysis.
Studies have demonstrated that automated SPE and tandem MS without HPLC columns can successfully quantify drugs at the picogram level, highlighting the sensitivity and efficiency achievable with modern SPE approaches.
LC-MS Integration with SPE for Metabolite Analysis
Sample Compatibility and Sensitivity Enhancement
The integration of SPE with liquid chromatography-mass spectrometry (LC-MS) represents a powerful combination for metabolite analysis. SPE provides the necessary sample clean-up to prevent matrix effects that can compromise LC-MS sensitivity and accuracy. By removing interfering compounds, SPE helps minimize ion suppression or enhancement effects that can significantly impact quantitative results.
Research has shown that SPE-mediated sample preparation significantly increases GC and LC column life while reducing downtime on sensitive instruments like LC-MS systems for source cleaning. The clean extracts produced by SPE allow for more reliable and reproducible LC-MS analysis, particularly at low concentration levels typical of metabolite studies.
On-line SPE-LC-MS Configurations
For maximum efficiency and sensitivity, on-line SPE-LC-MS configurations have been developed. These systems integrate SPE directly with the analytical instrumentation, allowing for automated sample processing and analysis. On-line approaches are particularly valuable for high-throughput applications and for analyzing unstable metabolites that might degrade during offline processing.
Studies have demonstrated successful on-line SPE-LC-MS applications for various analytes, including the determination of lipophilic antioxidants in blood plasma and the screening of polar pesticides in environmental waters. These configurations offer advantages in terms of reduced sample handling, improved reproducibility, and enhanced sensitivity.
Method Validation and Quality Control
When integrating SPE with LC-MS for metabolite analysis, rigorous method validation is essential. This includes assessment of extraction efficiency, matrix effects, precision, accuracy, and stability. Quality control samples should be processed alongside study samples to ensure method performance throughout analytical runs.
As noted in SPE methodology literature, lot-to-lot reproducibility of SPE cartridges is an important consideration for method validation. Studies have investigated the reproducibility of mixed-mode SPE columns in the extraction of drugs from whole blood, highlighting the need for consistent sorbent performance in regulated environments.
Future Directions and Considerations
The future of SPE in metabolite analysis continues to evolve with technological advancements. Miniaturization of SPE devices, development of new sorbent materials with enhanced selectivity, and improved automation capabilities are driving innovation in this field. Additionally, the investigation of extraction behavior for new drug metabolites remains an important area of research, particularly as new therapeutic agents are introduced to the market.
For laboratories considering SPE implementation for metabolite analysis, several key factors should be considered:
- Sorbent Selection: Choose SPE phases appropriate for the chemical properties of target metabolites
- Method Optimization: Develop and validate SPE methods specific to your analytes and matrices
- Automation Potential: Consider automated SPE systems for improved throughput and reproducibility
- Integration with Analytical Systems: Ensure compatibility between SPE protocols and downstream analytical instrumentation
- Quality Assurance: Implement appropriate controls and validation procedures
As the demands on analytical laboratories continue to increase—with requirements for lower detection limits, higher throughput, and more comprehensive metabolite profiling—SPE remains a critical technology for meeting these challenges in drug metabolite analysis.