Overview of Antibiotic Contamination in Food
Antibiotic residues in food products represent a significant public health concern with far-reaching implications for consumer safety and regulatory compliance. The use of antibiotics in livestock production for therapeutic treatment, disease prevention, and growth promotion has led to the potential presence of these compounds in meat, dairy products, eggs, and fish. According to research published in the Journal of Chromatography, liquid chromatographic analysis of antibacterial drug residues in food products of animal origin has revealed that antibiotic contamination can occur at various stages of food production, from farm to table.
The persistence of antibiotic residues in food matrices creates multiple challenges: potential allergic reactions in sensitive individuals, development of antibiotic-resistant bacteria, disruption of human gut microbiota, and interference with food fermentation processes. The complexity of food matrices—ranging from high-fat content in meat and dairy to protein-rich tissues—makes detection particularly challenging, necessitating sophisticated sample preparation techniques before analytical determination.
Common Antibiotic Classes in Livestock
Veterinary antibiotics encompass several major chemical classes, each with distinct physicochemical properties that influence their extraction and detection:
Sulfonamides
These broad-spectrum antibacterial agents are among the most commonly monitored residues. Studies have demonstrated their presence in various food matrices, including fish, honey, and animal tissues. Research by Horie et al. (1991) in the Journal of Chromatography highlighted simultaneous determination of residual synthetic antibacterials in fish using high-performance liquid chromatography, emphasizing the need for effective sample cleanup.
Tetracyclines
These compounds exhibit complexation behavior with metal ions and proteins, creating analytical challenges. Their amphoteric nature (containing both acidic and basic functional groups) requires careful pH control during extraction procedures.
β-Lactams (Penicillins and Cephalosporins)
Noted for their instability and reactivity, these antibiotics often require specialized extraction conditions to prevent degradation during sample preparation.
Macrolides and Aminoglycosides
These polar to moderately polar compounds present extraction challenges due to their hydrophilic nature and potential for strong matrix interactions.
Fluoroquinolones
These synthetic antibiotics exhibit zwitterionic properties, making them amenable to mixed-mode extraction approaches that combine reversed-phase and ion-exchange mechanisms.
Challenges in Detecting Trace Residues
The analytical determination of antibiotic residues in food samples presents multiple technical hurdles that must be addressed through optimized sample preparation:
Matrix Complexity
Food samples contain numerous interfering compounds—proteins, lipids, carbohydrates, pigments, and endogenous metabolites—that can co-extract with target analytes and interfere with detection. As noted in the Handbook of Sorbent Extraction Technology, milk, butter, and cheese present particularly challenging matrices due to their high fat and protein content and viscous or solid nature.
Low Concentration Levels
Regulatory limits for antibiotic residues typically range from parts-per-billion (ppb) to parts-per-million (ppm) levels, necessitating both effective cleanup and concentration capabilities. The work of Unruh et al. (1990) demonstrated solid-phase extraction of sulfamethazine in milk with quantitation at low ppb levels, highlighting the sensitivity requirements.
Chemical Diversity
The wide range of antibiotic structures—from polar, water-soluble compounds to lipophilic molecules—requires extraction methods capable of handling diverse physicochemical properties within a single analytical run.
Regulatory Compliance
Monitoring programs require methods that provide adequate recovery, precision, and accuracy to meet international standards such as those established by Codex Alimentarius, FDA, and EU regulations.
Role of SPE Cleanup Before LC-MS Analysis
Solid-phase extraction serves as a critical sample preparation step in antibiotic residue analysis, offering several advantages over traditional liquid-liquid extraction methods. As described in The Secrets of Solid Phase Extraction for Sample Preparation, SPE provides improved throughput through parallel processing, decreased organic solvent usage, higher and more reproducible recoveries, cleaner extracts, and elimination of emulsion formation.
Matrix Removal and Analyte Enrichment
SPE effectively removes interfering matrix components while concentrating target antibiotics, enhancing signal-to-noise ratios in subsequent LC-MS analysis. The technique’s tunable selectivity—achieved through careful choice of sorbent chemistry and solvent conditions—allows for optimization of recovery for specific antibiotic classes.
Method Development Strategy
Effective SPE method development for antibiotic analysis involves:
- Characterizing analyte properties (structure, pKa, polarity, functional groups)
- Understanding sample matrix composition and potential interferences
- Selecting appropriate sorbent chemistry based on analyte characteristics
- Optimizing loading, washing, and elution conditions
- Validating method performance against regulatory requirements
Sorbent Selection Considerations
Different SPE sorbents offer distinct advantages for antibiotic extraction:
- Reversed-phase sorbents (C18, C8): Effective for lipophilic antibiotics
- Mixed-mode sorbents: Combine reversed-phase and ion-exchange mechanisms for compounds with ionizable groups
- Hydrophilic-lipophilic balanced (HLB) polymers: Retain a broad range of compounds regardless of pH
- Ion-exchange sorbents: Selective for acidic or basic antibiotics at appropriate pH conditions
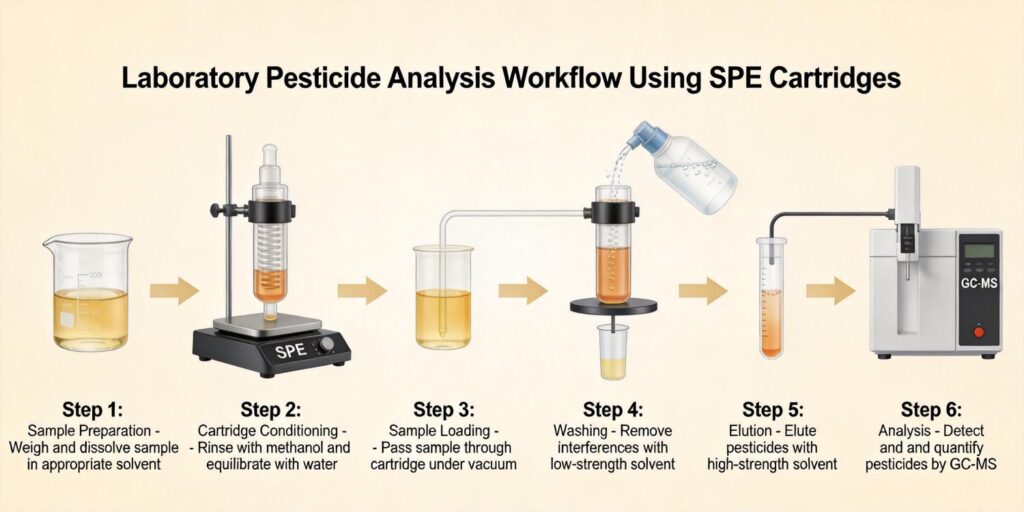
Example Workflow Using HLB Cartridges
Hydrophilic-lipophilic balanced (HLB) cartridges have emerged as particularly effective tools for multi-class antibiotic residue analysis due to their water-wettable polymer structure that provides balanced retention of both polar and non-polar compounds. The following workflow demonstrates their application in food analysis:
Sample Preparation
Begin with homogenization of the food sample (meat, fish, dairy) followed by extraction with appropriate solvents. For many antibiotic classes, acetonitrile or methanol with acidification (formic or acetic acid) provides effective extraction while precipitating proteins.
SPE Procedure
- Conditioning: Activate the HLB cartridge with methanol followed by water or buffer
- Loading: Apply the sample extract at controlled flow rates (1-3 mL/min)
- Washing: Remove weakly retained interferences with water or low-percentage organic solvents
- Drying: Remove residual water to prevent dilution of elution solvent
- Elution: Recover antibiotics with appropriate organic solvents (methanol, acetonitrile, often with acid or base modifiers)
Concentration and Reconstitution
Evaporate the eluate to dryness and reconstitute in mobile phase compatible with LC-MS analysis, achieving both cleanup and concentration factors of 10-100x.
Method Validation
As demonstrated in studies of sulfonamide residues, HLB-based methods can achieve recoveries exceeding 90% with relative standard deviations below 10% for multiple antibiotic classes simultaneously. The work of Horie et al. on simultaneous determination of residual synthetic antibacterials in fish exemplifies the multi-residue capability of well-optimized SPE methods.
Regulatory Limits and Monitoring Programs
International regulatory frameworks establish maximum residue limits (MRLs) for antibiotics in food products, driving the need for sensitive and reliable analytical methods.
Global Standards
Major regulatory bodies including the U.S. Food and Drug Administration (FDA), European Food Safety Authority (EFSA), and Codex Alimentarius Commission establish MRLs based on toxicological assessments, considering factors such as acceptable daily intake, food consumption patterns, and safety margins.
Monitoring Programs
National residue monitoring programs employ surveillance testing to ensure compliance with established limits. These programs typically utilize multi-residue methods capable of detecting numerous antibiotic classes in various food matrices. The development of such methods, as described in chromatographic analyses of antibiotic materials in food, represents a significant analytical achievement.
Analytical Requirements
Regulatory methods must demonstrate:
- Specificity: Ability to distinguish target antibiotics from matrix interferences
- Sensitivity: Detection at or below established MRLs
- Accuracy and Precision: Recovery within acceptable ranges with minimal variability
- Ruggedness: Consistent performance across different laboratories and analysts
- Throughput: Capacity to handle large sample volumes in monitoring programs
Future Directions
Advancements in SPE technology continue to enhance antibiotic residue monitoring. Automated SPE systems, 96-well plate formats, and on-line SPE-LC-MS configurations improve throughput and reproducibility. New sorbent chemistries, including those specifically designed for veterinary drug analysis, offer improved selectivity and cleaner extracts. As monitoring requirements become more stringent and the number of regulated compounds increases, SPE will remain an essential tool in ensuring food safety through effective antibiotic residue detection.
The integration of optimized SPE methods with sensitive detection techniques like LC-MS/MS provides regulatory laboratories with powerful tools for protecting public health. By effectively addressing the challenges of matrix complexity, low concentration levels, and chemical diversity, SPE enables reliable monitoring of antibiotic residues in the global food supply.