1. Major Mycotoxins Monitored in Grain Testing
Grain safety testing programs worldwide focus on several critical mycotoxins that pose significant health risks to humans and animals. The primary mycotoxins monitored in grain samples include aflatoxins (AFLA B1, B2, G1, G2), ochratoxin A, fumonisins (B1, B2), deoxynivalenol (DON), zearalenone, and T-2/HT-2 toxins. Among these, aflatoxin B1 is classified as a Group 1 human carcinogen by the International Agency for Research on Cancer (IARC), with maximum limits established at 5 μg/kg for AFLA B1 and 10 μg/kg for total aflatoxins in maize products in the European Union.
Recent research by Massarolo et al. (2018) demonstrated successful simultaneous determination of aflatoxins G2, G1, B2, and B1 in cornmeal using matrix solid-phase dispersion (MSPD) extraction coupled with HPLC-fluorescence detection. Their study achieved limits of detection ranging from 0.01 to 0.04 ng/g and limits of quantification from 0.02 to 0.1 ng/g, well below regulatory thresholds. The method showed recoveries between 85.7% and 114.8%, meeting European Commission criteria of 70-110% for concentrations between 1-10 μg/kg.
2. Sample Extraction from Grain Matrices
Effective sample preparation begins with proper homogenization of grain samples, typically using grinding to achieve consistent particle size distribution. Research indicates that particle size significantly affects extraction efficiency, with coarse and medium grind cornmeals showing better recoveries than fine cornmeals. Massarolo et al. found that medium cornmeal (particles between 0.355-0.25 mm) provided optimal extraction conditions for aflatoxins.
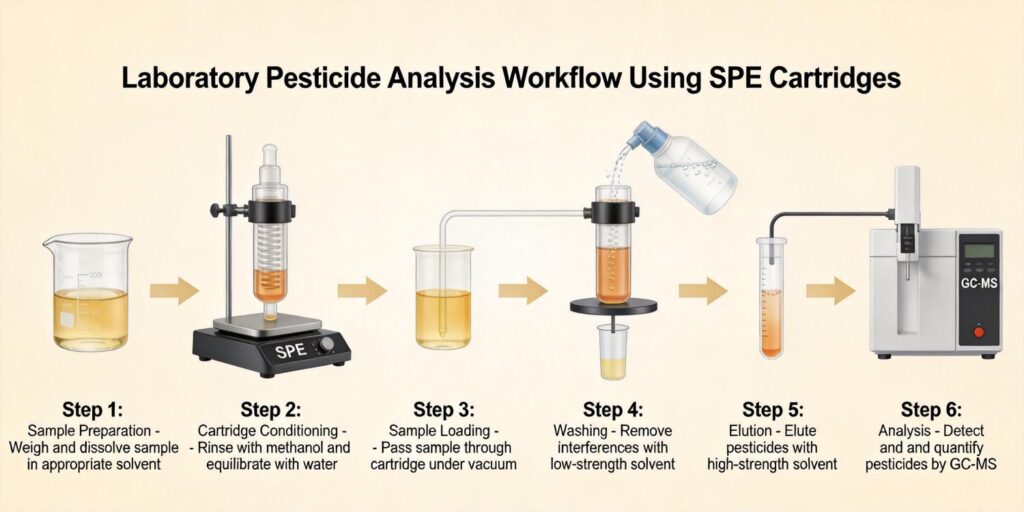
Matrix solid-phase dispersion (MSPD) has emerged as a powerful extraction technique for mycotoxins in cereals. This approach combines sample disruption, extraction, and cleanup in a single step, reducing solvent consumption and analysis time. In MSPD, the sample is blended with a solid support material (typically C18 or celite) using a mortar and pestle or vortex mixer, creating a homogeneous mixture that is then packed into a column for elution.
The vortex-assisted MSPD method developed by Massarolo et al. used 1 g of cornmeal blended with 25 mg of C18 solid support, followed by elution with 10 mL of acetonitrile/methanol (50:50, v/v). This approach eliminated the need for column packing and reduced variations associated with traditional MSPD procedures.
3. SPE Sorbent Choices for Mycotoxin Cleanup
Selecting appropriate solid-phase extraction sorbents is crucial for effective mycotoxin cleanup from complex grain matrices. The choice depends on the polarity of target mycotoxins and the nature of matrix interferences. For aflatoxin analysis in cereals, C18 reversed-phase sorbents are most commonly used due to their lipophilic characteristics, which allow good disruption, dispersion, and retention of lipophilic species.
Research comparing different solid supports for aflatoxin extraction from cornmeal found that C18 provided superior recoveries compared to celite. Massarolo et al. optimized their MSPD method using a central composite design and determined that 25 mg of C18 provided optimal recoveries for all four aflatoxins tested. The study also investigated mixed sorbent systems, finding that pure C18 outperformed C18:celite mixtures for aflatoxin extraction.
For multi-mycotoxin analysis, mixed-mode sorbents combining reversed-phase and ion-exchange functionalities offer advantages. These sorbents can simultaneously extract mycotoxins with varying polarities while removing interfering matrix components. The selection should consider the specific mycotoxin classes being analyzed and the grain matrix composition.
4. Washing Strategies to Remove Pigments and Fats
Grain matrices contain numerous interfering compounds including pigments (chlorophyll, carotenoids), lipids, proteins, and sugars that can co-extract with mycotoxins and interfere with chromatographic analysis. Effective washing strategies are essential to remove these interferences while retaining target analytes.
For aflatoxin analysis in cornmeal, research has shown that proteins and sugars are significant interferers. Massarolo et al. found strong negative correlations between protein content and AFLA G2 recovery (r = -0.99) and between sugars and AFLA G1 recovery (r = -0.99). To address these interferences, they modified their extraction method by increasing C18 amount from 25 mg to 100 mg and adding 0.25 g anhydrous sodium sulfate, which improved recoveries for affected aflatoxins.
Typical washing solvents for mycotoxin SPE include water, aqueous methanol (5-20%), or buffer solutions. The washing step should be optimized to remove polar interferences without eluting target mycotoxins. For reversed-phase SPE, water or low-percentage methanol washes effectively remove polar pigments and sugars, while stronger washes with higher organic content may be needed for more complex matrices.
5. Elution Optimization for LC-MS Detection
Elution optimization is critical for achieving high recoveries and compatibility with downstream analytical techniques, particularly LC-MS/MS. For aflatoxins in grain matrices, acetonitrile-based eluents generally provide better recoveries than methanol-based systems. Massarolo et al. found that acetonitrile/methanol (50:50, v/v) provided optimal elution for aflatoxins from C18 sorbents.
The study employed HPLC with fluorescence detection and post-column photochemical derivatization, achieving excellent separation of aflatoxins G2, G1, B2, and B1 in 14 minutes using a mobile phase of water/acetonitrile/methanol (62:14:24, v/v/v) in isocratic mode. For LC-MS applications, elution solvents should be compatible with the ionization technique and provide adequate analyte solubility.
Elution volume optimization is equally important. While traditional methods may use 20-30 mL of elution solvent, modern approaches aim to minimize solvent consumption. The optimized MSPD method used only 10 mL of elution solvent, reducing waste and improving concentration factors. For trace analysis, eluates are typically evaporated and reconstituted in smaller volumes compatible with LC-MS injection.
6. Method Validation Parameters
Comprehensive method validation is essential for regulatory compliance and reliable mycotoxin analysis. Key validation parameters include accuracy (recovery), precision (repeatability and reproducibility), linearity, limit of detection (LOD), limit of quantification (LOQ), and matrix effects.
Massarolo et al. validated their MSPD method according to SANTE/11945/2015 guidelines, achieving recoveries of 85.7-114.8% across three fortification levels. The method demonstrated excellent precision with relative standard deviations (RSDs) below 20%, meeting European Commission criteria. LODs ranged from 0.01-0.04 ng/g and LOQs from 0.02-0.1 ng/g, sufficient for regulatory compliance monitoring.
Matrix effects were evaluated by comparing calibration slopes in solvent and matrix-matched standards. The study found matrix effects of approximately 20% for AFLAs G2 and G1 and 11% for B2 and B1, within acceptable limits for trace analysis according to SANTE guidelines. Method applicability was demonstrated across different cornmeal types (coarse, medium, fine) and processing methods (dry-milling, wet-milling).
7. Regulatory Testing Requirements
Mycotoxin testing in grains must comply with international regulatory frameworks that establish maximum limits and analytical requirements. The European Union’s Commission Regulation (EC) No 1881/2006 sets maximum levels for various mycotoxins in foodstuffs, including 5 μg/kg for AFLA B1 and 10 μg/kg for total aflatoxins in maize products. Brazil’s ANVISA establishes a limit of 20 μg/kg for total aflatoxins in corn.
Regulatory methods must meet specific performance criteria. For aflatoxin concentrations below 1 μg/kg, recoveries should range between 50-120%, while for concentrations between 1-10 μg/kg, recoveries should be 70-110%. Precision requirements typically specify RSDs below 20% for the concentrations used in repeatability testing.
Comparative studies have shown that MSPD methods can achieve performance comparable to traditional immunoaffinity column cleanup methods while offering advantages in solvent consumption and sample throughput. Massarolo et al. demonstrated that their MSPD method consumed only 10 mL of solvent compared to 70-150 mL in standard methods, with comparable or better LODs and recoveries.
Conclusion
Effective SPE workflows for trace mycotoxin analysis in grain samples require careful consideration of extraction techniques, sorbent selection, washing strategies, and elution optimization. The development of matrix solid-phase dispersion methods represents significant progress toward greener analytical approaches with reduced solvent consumption and improved efficiency. As regulatory requirements become more stringent and analytical techniques more sensitive, continued optimization of SPE workflows will remain essential for ensuring grain safety and compliance with international standards.
References:
Massarolo, K.C., Ferreira, C.F.J., Kupski, L., & Badiale-Furlong, E. (2018). Optimization of Matrix Solid-Phase Dispersion Method for Extraction of Aflatoxins from Cornmeal. Food Analytical Methods, 11, 1-10.
European Commission. (2006). Commission Regulation (EC) No 1881/2006 setting maximum levels for certain contaminants in foodstuffs.
SANTE. (2016). Analytical quality control and method validation procedures for pesticide residues analysis.