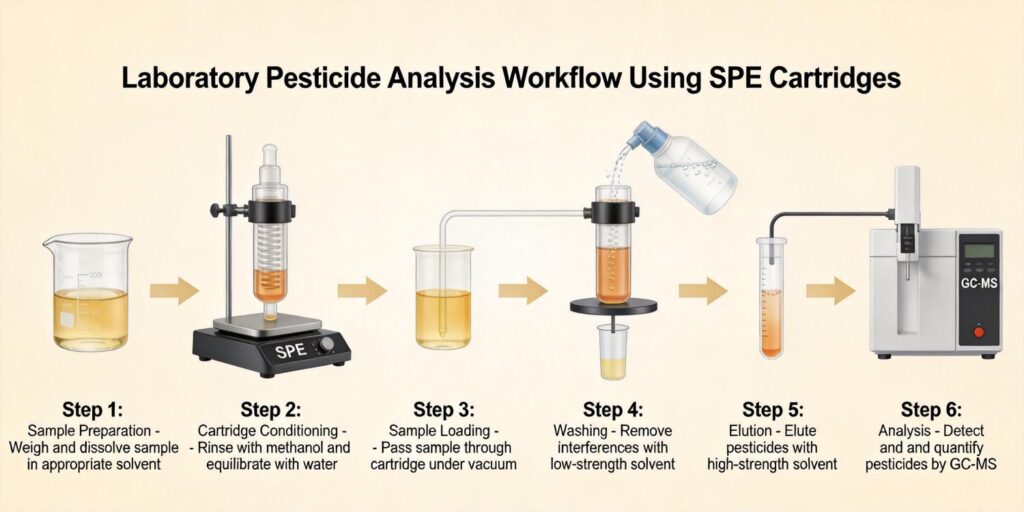
Fermented Food Matrices and Organic Acid Composition
Fermented foods represent a diverse category of complex matrices that include dairy products (yogurt, cheese), vegetables (sauerkraut, kimchi), beverages (wine, beer), and soy products (miso, tempeh). These foods undergo microbial transformation where organic acids play crucial roles in flavor development, preservation, and nutritional quality. The primary organic acids found in fermented foods include lactic acid, acetic acid, citric acid, malic acid, tartaric acid, and succinic acid, each contributing distinct sensory properties and analytical challenges.
According to Simpson and Wynne (2000), wine analysis requires examination of both volatile species responsible for bouquet and sugar acids that contribute to flavor. Solid-supported liquid-liquid extraction (LLE) and solid-phase extraction (SPE) have been extensively used for this purpose, with SPE offering advantages in class fractionation into acid, base, and neutral fractions. The ability to extract under mild pH conditions limits decomposition or rearrangement of labile compounds, which is particularly important for fermented food analysis where acid-sensitive compounds may be present.
Extraction Using Water or Dilute Acid Solution
The initial extraction step for organic acids from fermented foods typically involves using water or dilute acid solutions. For most fermented matrices, water extraction at room temperature effectively solubilizes water-soluble organic acids while minimizing co-extraction of interfering compounds. Dilute acid solutions (typically 0.1-1% formic or acetic acid) can enhance extraction efficiency for certain organic acids by maintaining acidic conditions that prevent dissociation and improve solubility.
Research has shown that ethanol can serve as an effective extractant with relatively low toxicity exposure for operators. In studies examining fat-soluble vitamins in feed samples, ethanol was selected as the extractant due to its effectiveness in solubilizing target compounds while minimizing co-extraction of interfering matrix components. The choice between water and dilute acid solutions depends on the specific fermented food matrix and target organic acid profile.
Removal of Solids via Centrifugation and Filtration
Following extraction, fermented food extracts typically contain suspended solids, proteins, and other particulate matter that must be removed prior to SPE. Centrifugation at 3,000-5,000 × g for 10-15 minutes effectively separates solids from the liquid extract. Subsequent filtration through 0.45 μm or 0.22 μm membrane filters ensures complete removal of fine particulates that could clog SPE cartridges or interfere with subsequent chromatographic analysis.
In complex matrices like cheese or fermented meat products, additional sample preparation may be required. De Jong and Badings (1990) demonstrated that cheese samples could be ground with anhydrous salt and sulfuric acid, followed by ether/heptane extraction, to prepare samples for SPE analysis of fatty acids. For milk samples, ethanol and sulfuric acid addition followed by similar extraction procedures proved effective.
Selection of MAX SPE Cartridge for Acidic Compound Retention
The Mixed-mode Anion eXchange (MAX) SPE cartridge represents the optimal choice for organic acid extraction from fermented foods. MAX cartridges combine reversed-phase retention with anion exchange functionality, providing dual retention mechanisms that ensure efficient capture of acidic compounds. According to Waters documentation, MAX sorbents have a tightly controlled ion-exchange capacity of 0.25 meq/g, ensuring reproducible SPE protocols for extraction of acidic compounds and metabolites.
The mixed-mode retention mechanism is particularly advantageous for fermented food analysis because it allows retention of organic acids through both hydrophobic interactions (with the polymeric backbone) and ionic interactions (with the anion exchange sites). This dual mechanism provides superior selectivity compared to traditional reversed-phase sorbents, especially for polar organic acids that might otherwise show poor retention on C18 or C8 phases.
Why MAX Outperforms Other SPE Options
Traditional reversed-phase sorbents like C18 often struggle with highly polar organic acids common in fermented foods. Strong anion exchange (SAX) sorbents can retain these acids effectively but may require harsh elution conditions. MAX cartridges offer the ideal compromise: they retain organic acids at neutral or basic pH through anion exchange while providing additional hydrophobic retention. The water-wettable polymeric sorbent is stable across pH 0-14, making method development straightforward and robust.
Conditioning with Methanol and Water
Proper conditioning of MAX cartridges is essential for optimal performance. The standard conditioning protocol involves:
- Methanol conditioning: 2-3 mL of methanol to solvate the polymeric sorbent and ensure complete wetting of the hydrophobic surface
- Water equilibration: 2-3 mL of deionized water or buffer to remove methanol and prepare the sorbent for aqueous sample loading
This two-step conditioning process activates both the reversed-phase and ion-exchange functionalities of the MAX sorbent. The methanol step ensures complete wetting of the hydrophobic polymer, while the water step establishes the appropriate ionic environment for anion exchange interactions. For fermented food extracts, using the same pH buffer for conditioning as will be used for sample loading (typically pH 7-8 for organic acid retention) helps maintain consistent retention characteristics.
Loading Diluted Extract onto Cartridge
Sample loading represents a critical step where method parameters significantly impact recovery and selectivity. For MAX cartridges, organic acids should be loaded in their ionized form (typically at pH 7-8) to maximize anion exchange retention. The sample should be diluted sufficiently to reduce organic solvent content (typically to less than 10% organic) and adjust ionic strength to optimal levels.
Research on SPE optimization for vitamins demonstrated the importance of loading solvent composition. When examining retention of fat-soluble vitamins on HLB columns, various ethanol concentrations (15-95%) were evaluated. The results indicated optimal retention at 65% ethanol, highlighting how solvent composition during loading significantly affects analyte retention. For MAX cartridges and organic acids, maintaining appropriate pH and minimizing organic content during loading ensures effective anion exchange retention.
Washing with Water to Remove Sugars
Fermented foods often contain significant amounts of sugars (glucose, fructose, lactose) and other polar, neutral compounds that can interfere with organic acid analysis. A water wash step effectively removes these interferences while retaining ionized organic acids on the MAX sorbent. Typically, 2-3 mL of deionized water or a mild buffer (pH 7-8) suffices to elute sugars and other polar neutrals.
In wine analysis, SPE has been used to extract pigments (anthocyanins) while leaving sugars in the effluent, demonstrating effective class separation. This crude fractionation could be extended by passing wine through an anion exchanger to trap wine acids, as demonstrated by Saito et al. (1989) in the clean-up of soft drinks during aspartame degradation product analysis.
Optimizing Wash Conditions
The wash step should be sufficiently stringent to remove interferences but gentle enough to prevent premature elution of target organic acids. For MAX cartridges, water or low-ionic-strength buffers at neutral to slightly basic pH provide optimal washing conditions. Adding 5-10% methanol to the wash solvent can help remove moderately polar interferences without compromising organic acid retention, though this requires careful optimization for specific applications.
Elution with Acidic Methanol
Organic acids are efficiently eluted from MAX cartridges using acidic methanol, typically methanol containing 2-5% formic acid or acetic acid. The acid serves two purposes: it protonates the anion exchange sites, disrupting ionic interactions, and it protonates the organic acids themselves, converting them to their neutral forms that show reduced retention on the reversed-phase component.
The elution volume should be minimized to concentrate analytes while ensuring complete recovery. Typically, 2-3 mL of acidic methanol provides quantitative elution for most organic acids from fermented foods. For particularly strongly retained acids (those with multiple carboxyl groups or aromatic systems), increasing acid concentration to 5% or including a small percentage of stronger acid (like TFA) may improve recovery.
Quantitative Analysis Using HPLC or LC-MS
Following SPE extraction and concentration, organic acids from fermented foods are typically analyzed by reversed-phase HPLC with UV detection (210-220 nm for carboxyl groups) or by LC-MS for enhanced sensitivity and specificity. HPLC methods commonly employ C18 columns with acidic mobile phases (typically 0.1% formic acid or phosphoric acid in water) and gradient elution with increasing acetonitrile or methanol.
For complex fermented food matrices, LC-MS provides superior selectivity, particularly when analyzing trace organic acids or distinguishing between isomeric compounds. Mass detection allows specific monitoring of target acids even in the presence of co-eluting interferences. Simpson and Wynne (2000) note that SPE extracts are easily amenable to subsequent GC- and HPLC-analysis, with chromatograms showing minimal interference from endogenous matrix components when proper SPE protocols are followed.
Method Validation Considerations
Validation of the complete SPE-HPLC/LC-MS method should include assessment of linearity, sensitivity, precision, and accuracy across the expected concentration range for target organic acids in fermented foods. Recovery studies using spiked samples provide crucial data on method performance, while comparison with reference methods or standard addition approaches confirms accuracy.
Research on fat-soluble vitamin analysis demonstrated method validation with respect to linearity and sensitivity, plus precision and accuracy assessment. Recovery studies showed values ranging from 87.6% to 129.6% for different vitamins at various spiking levels, with coefficients of variation typically below 10%, indicating robust method performance.
Conclusion: Optimizing SPE for Fermented Food Analysis
The MAX SPE cartridge workflow for organic acid extraction from fermented foods represents a robust, selective approach that addresses the unique challenges posed by these complex matrices. By leveraging mixed-mode retention mechanisms, MAX cartridges provide superior recovery and cleanliness compared to traditional SPE approaches. Proper attention to extraction conditions, sample preparation, and chromatographic analysis ensures accurate quantification of organic acids that define the sensory and quality characteristics of fermented foods.
For laboratories analyzing fermented food products, implementing this MAX-based SPE workflow offers several advantages: improved selectivity for organic acids, reduced matrix interference, compatibility with both HPLC and LC-MS detection, and the ability to handle diverse fermented food matrices with minimal method modification. As Simpson and Wynne (2000) concluded, SPE is a very useful tool for sample workup, isolation, and concentration, providing extracts that are easily amenable to subsequent chromatographic analysis with minimal interference from endogenous matrix components.