Regulatory Monitoring of Veterinary Drugs
Veterinary drug residue monitoring in meat products represents a critical public health safeguard implemented by regulatory agencies worldwide. The detection of antibacterial drugs, sulfonamides, and other therapeutic agents in animal tissues requires robust analytical methodologies that can achieve detection limits at parts-per-billion (ppB) levels. As noted in the literature, “antibacterial drug residues in food products of animal origin” require sensitive detection methods to ensure consumer safety and regulatory compliance.
Regulatory frameworks such as those established by the FDA, EU Commission, and Codex Alimentarius establish maximum residue limits (MRLs) for various veterinary drugs in meat, poultry, and fish. These regulations necessitate analytical methods capable of detecting compounds like sulfamethazine, tetracyclines, and anabolic steroids at concentrations as low as 1-10 μg/kg. The complexity of meat matrices, rich in proteins, lipids, and pigments, presents significant challenges for residue analysis that solid-phase extraction (SPE) effectively addresses.
Homogenization of Meat Samples
The initial step in veterinary drug residue analysis involves proper sample homogenization to ensure representative sampling and efficient extraction. Meat tissue, whether from cattle, pigs, poultry, or fish, requires mechanical disruption to release analytes from cellular structures. As described in veterinary applications, “adipose or muscle tissue is usually analyzed with the intent to determine the health effects of consumption of that tissue and, as such, focuses on identification of pesticides or drug residues.”
Effective homogenization typically involves blending or grinding the meat sample with appropriate extraction solvents. For fish tissue analysis, researchers have employed slurrying buffers containing metaphosphoric acid/methanol mixtures (3:2 v/v) to deproteinize samples before filtration and concentration. The homogenization process must balance complete tissue disruption with minimal analyte degradation, particularly for heat-sensitive compounds. Proper homogenization ensures that subsequent extraction steps can efficiently recover target analytes from the complex meat matrix.
Solvent Extraction with Acetonitrile
Following homogenization, solvent extraction serves to separate veterinary drug residues from the bulk meat matrix. Acetonitrile has emerged as the preferred extraction solvent for many veterinary drug classes due to its excellent solubilizing properties and compatibility with subsequent SPE cleanup steps. Acetonitrile effectively precipitates proteins while extracting a broad range of veterinary drugs including sulfonamides, tetracyclines, and β-lactams.
The extraction process typically involves mixing homogenized meat with acetonitrile, often with added salts (such as magnesium sulfate) to induce phase separation through the QuEChERS (Quick, Easy, Cheap, Effective, Rugged, Safe) approach. This method partitions analytes into the acetonitrile layer while leaving proteins, sugars, and some pigments in the aqueous phase. As noted in veterinary drug analysis literature, “the high protein and lipid content of these samples often makes them prone to emulsification during liquid-liquid extraction,” making SPE an attractive alternative for cleaner extracts.
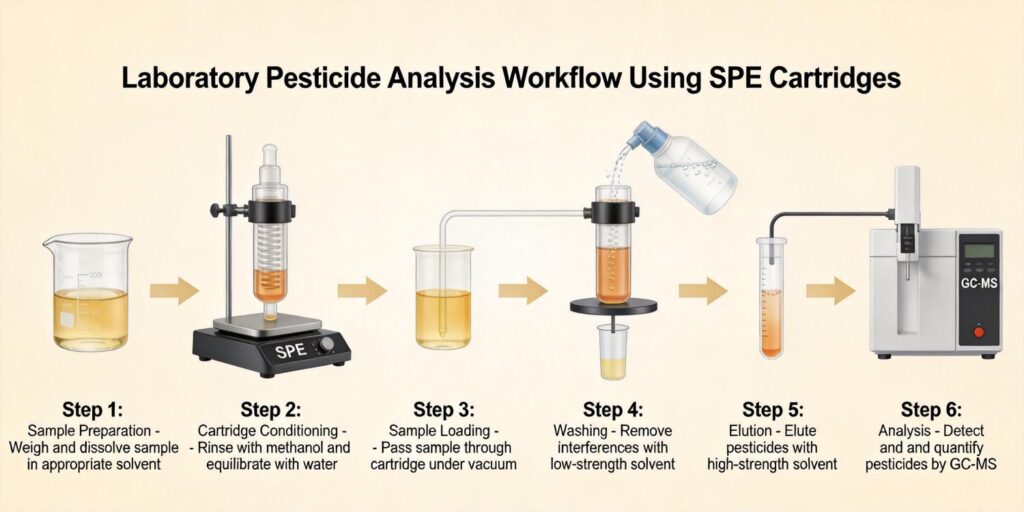
SPE Cleanup Using HLB Cartridges
Solid-phase extraction with hydrophilic-lipophilic balanced (HLB) cartridges represents a critical cleanup step in veterinary drug residue analysis. HLB sorbents, composed of poly(divinylbenzene-co-N-vinylpyrrolidone), provide dual retention mechanisms through both hydrophilic and lipophilic interactions, making them ideal for extracting a wide range of veterinary drugs with varying polarities.
As demonstrated in feed analysis applications, “Oasis HLB cartridges containing poly divinylbenzene-co-N-vinylpyrrolidone sorbents exhibit both hydrophilic and lipophilic retention characteristics, playing a valid role in the extraction of medium-polar and non-polar organic compounds from mixtures of water and organic solvent.” For meat extracts, HLB cartridges effectively retain veterinary drugs while allowing many matrix interferences to pass through during the wash steps.
The SPE procedure typically involves conditioning the HLB cartridge with methanol followed by water or buffer, loading the acetonitrile extract (often diluted with water to reduce organic content), washing with appropriate solvents, and finally eluting the target analytes. This process significantly reduces matrix effects that can interfere with subsequent LC-MS analysis.
Optimizing HLB Cartridge Performance
Proper optimization of HLB cartridge conditions is essential for maximizing veterinary drug recovery. Research on fat-soluble vitamin extraction from feeds provides valuable insights applicable to veterinary drug analysis. Studies have shown that “all vitamins can be held on the cartridge under 45%, 55%, and 65% ethanol solvent,” with 65% ethanol often selected for optimal retention. For veterinary drugs in meat extracts, similar optimization of loading solvent composition (typically water:acetonitrile mixtures) ensures complete analyte retention while allowing matrix components to pass through.
Removal of Fats and Pigments During Wash Step
Meat extracts contain significant amounts of lipids, cholesterol, and pigments that can interfere with analytical detection. The wash step in SPE procedures serves to remove these interferences while retaining target veterinary drugs. HLB cartridges allow selective washing with solvents that remove non-polar interferences without eluting the analytes of interest.
Common wash solvents include water with low percentages of methanol or acetonitrile (typically 5-10%), which effectively remove polar matrix components while retaining veterinary drugs on the HLB sorbent. As noted in veterinary applications, “the cartridge is washed with water to remove salts and other soluble material and with hexane to both remove some non-polar material and aid in drying.” For meat extracts, sequential washing with aqueous solutions followed by non-polar solvents like hexane or ethyl acetate effectively removes fats and pigments.
The selective removal of matrix components is particularly important for LC-MS analysis, where ion suppression from co-eluting compounds can significantly affect method sensitivity and accuracy. Proper wash optimization ensures clean extracts that produce reliable quantitative results at regulatory limits.
Elution Solvent Selection for LC-MS Analysis
Elution solvent selection represents a critical parameter in SPE method development for veterinary drug residue analysis. The elution solvent must completely recover target analytes while maintaining compatibility with subsequent LC-MS analysis. For HLB cartridges, methanol and acetonitrile, often with added acid or base modifiers, provide effective elution for most veterinary drugs.
Research on SPE optimization demonstrates that “several solvents with different polarities, such as tetrahydrofuran, acetonitrile, ethyl acetate, acetone, cyclohexane, ethanol, and methanol, were chosen as the eluent. Three volumes (1, 2, and 3 mL) were assayed. Taking into consideration the volatility, highest recovery, and smallest volume, 1 mL of ethanol was set as eluent.” For veterinary drugs in meat, methanol or acetonitrile with 2-5% formic acid or ammonium hydroxide often provides optimal elution efficiency.
The elution volume must balance complete analyte recovery with minimal dilution of the extract. Typically, 2-5 mL of elution solvent provides quantitative recovery while maintaining adequate concentration for sensitive detection. The eluate is often evaporated to dryness and reconstituted in mobile phase compatible with LC-MS analysis, typically water:methanol or water:acetonitrile mixtures with appropriate buffers.
pH Considerations in Elution
For ionizable veterinary drugs, pH adjustment during elution can significantly enhance recovery. Basic drugs (such as many antibiotics) elute more efficiently with alkaline eluents (methanol with 2-5% ammonium hydroxide), while acidic drugs require acidic conditions (methanol with 2-5% formic acid). Neutral veterinary drugs elute effectively with pure organic solvents. Understanding the pKa values of target analytes allows optimization of elution pH for maximum recovery.
Recovery Validation Strategy
Method validation for veterinary drug residue analysis requires comprehensive recovery studies to demonstrate analytical reliability. Recovery validation typically involves spiking blank meat matrices with known concentrations of target analytes at multiple levels (often at the regulatory limit, half the limit, and twice the limit), followed by complete sample preparation and analysis.
As demonstrated in tissue analysis studies, extraction yields for various drugs from spiked tissues show acceptable recoveries with relative standard deviations typically below 10%. For example, research on liver tissue analysis reported “extraction yields of twenty drugs extracted from liver tissue after enzymatic digestion” with spike levels of 2 μg/100 mg wet tissue, demonstrating the method’s capability for sensitive detection.
Recovery validation should include:
- Matrix-matched calibration: Using blank meat extracts spiked with known concentrations to account for matrix effects
- Internal standards: Employing stable isotope-labeled analogs of target analytes to correct for extraction efficiency variations
- Quality control samples: Including low, medium, and high concentration QC samples in each batch to monitor method performance
- Inter-laboratory comparisons: Participating in proficiency testing programs to ensure method accuracy and precision
Method Performance Characteristics
Validated methods for veterinary drug residue analysis in meat should demonstrate:
- Recovery: Typically 70-120% for most analytes
- Precision: Relative standard deviation ≤15% for repeatability and intermediate precision
- Linearity: Correlation coefficient (r²) ≥0.99 over the working range
- Limit of detection: Sufficiently below the regulatory MRL (typically 1/3 to 1/10 of MRL)
- Specificity: No interference from matrix components at the retention times of target analytes
The comprehensive SPE approach using HLB cartridges, when properly validated, provides reliable results for regulatory monitoring of veterinary drug residues in meat products. This methodology supports food safety programs worldwide by enabling sensitive, accurate detection of prohibited substances at concentrations relevant to public health protection.
For laboratories implementing veterinary drug residue analysis, Poseidon Scientific offers HLB SPE cartridges specifically designed for complex matrix applications. Our products provide consistent lot-to-lot reproducibility and excellent recovery characteristics for a wide range of veterinary drugs, supporting reliable analytical results for food safety monitoring programs.