Overview of Pesticide Residue Regulations and Detection Limits in Fruit Testing
Global food safety regulations have established stringent limits for pesticide residues in fruits, with maximum residue limits (MRLs) typically ranging from 0.01 to 10 mg/kg depending on the pesticide and fruit type. Regulatory bodies including the European Union, US FDA, and Codex Alimentarius Commission enforce these standards to ensure consumer safety. The Luke method, developed for multiresidue pesticide analysis, has evolved to meet increasingly demanding sensitivity requirements, with detection limits now reaching parts-per-billion (ppb) levels for many compounds.
Modern analytical techniques, particularly LC-MS/MS, enable simultaneous detection of hundreds of pesticide residues in a single run. As noted in comprehensive studies, “the method goals became more stringent” over time, requiring enhanced sample preparation techniques to achieve the necessary sensitivity and selectivity. The QuEChERS (Quick, Easy, Cheap, Effective, Rugged, and Safe) approach has revolutionized pesticide residue analysis, with studies demonstrating analysis of 229 pesticides with recoveries of 90-110% and RSDs <5%.
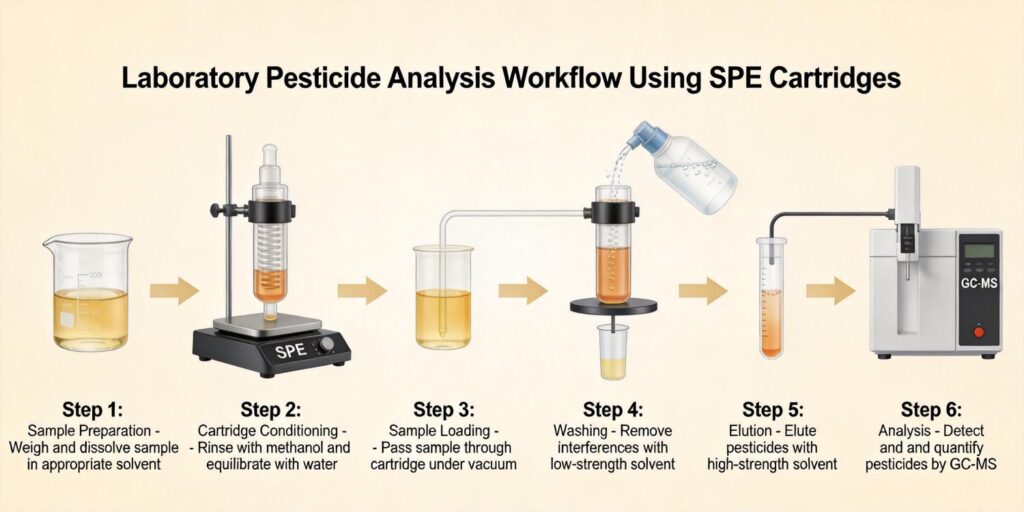
Sample Homogenization and Extraction Using Acetonitrile-Based QuEChERS Extraction
The QuEChERS method begins with thorough homogenization of fruit samples to ensure representative sampling. Typically, 10-15 grams of homogenized sample are weighed into a 50 mL centrifuge tube. The extraction phase involves adding 10 mL of acetonitrile, which serves as the primary extraction solvent due to its excellent pesticide solubility and ability to precipitate proteins and other matrix components.
According to Waters documentation, “QuEChERS methods offer a simple and straightforward sample preparation technique ideal for multi-residue analysis for pesticides, veterinary drugs, and mycotoxins in a wide variety of food and agricultural products.” The method typically includes addition of salts such as magnesium sulfate (MgSO₄) for dehydration and sodium chloride (NaCl) for phase separation. For AOAC methods, 1.5 g sodium acetate and 6 g MgSO₄ are commonly used, while CEN methods employ 1 g trisodium citrate dihydrate, 0.5 g disodium hydrogencitrate sesquihydrate, 1 g NaCl and 4 g MgSO₄.
The mixture is vigorously shaken for 1-2 minutes, then centrifuged to separate the organic phase from the aqueous layer and solid residues. This initial extraction efficiently transfers pesticides from the fruit matrix into the acetonitrile phase while leaving many interfering compounds behind.
Dilution of Extract to Reduce Organic Solvent Strength Before SPE Loading
Following QuEChERS extraction, the acetonitrile extract contains approximately 80-100% organic solvent, which is too strong for effective retention on HLB SPE cartridges. Proper dilution is critical for maximizing pesticide retention through reversed-phase interactions. Typically, the acetonitrile extract is diluted with water or aqueous buffer to achieve a final organic solvent concentration of 5-20%.
This dilution step serves multiple purposes: it reduces the solvent strength to allow proper analyte retention on the HLB sorbent, adjusts the sample volume for optimal loading conditions, and can help precipitate additional matrix components. The exact dilution factor depends on the specific fruit matrix and pesticide panel being analyzed, but a 1:4 to 1:10 dilution with water is commonly employed.
Conditioning HLB Cartridge with Methanol Followed by Water
Proper conditioning of HLB (Hydrophilic-Lipophilic Balance) cartridges is essential for achieving consistent and reproducible results. The conditioning sequence typically involves:
- Methanol conditioning: 3-5 mL of methanol is passed through the cartridge to wet the polymeric sorbent and remove any residual impurities from manufacturing. This step ensures complete activation of the reversed-phase retention sites.
- Water conditioning: 3-5 mL of deionized water is passed through the cartridge to remove methanol and create an aqueous environment compatible with the diluted fruit extract.
It’s crucial that the sorbent bed does not dry out between conditioning and sample loading, as this can create channels and reduce retention efficiency. The conditioning solvents should be allowed to flow through the cartridge by gravity or gentle vacuum, with approximately 1-2 mL remaining above the sorbent bed when sample loading begins.
Loading Diluted Fruit Extract and Retaining Pesticides via Reversed-Phase Interaction
The diluted fruit extract is loaded onto the conditioned HLB cartridge at a controlled flow rate, typically 1-5 mL/min. HLB sorbent, composed of a hydrophilic N-vinylpyrrolidone and lipophilic divinylbenzene copolymer, provides excellent retention for a wide range of pesticide compounds through reversed-phase mechanisms.
The polymeric structure of HLB offers several advantages for pesticide analysis:
- High capacity: Typically 5-15 mg/mL sorbent bed for moderately hydrophobic compounds
- pH stability: Effective performance across pH 0-14 range
- Wettability: Maintains retention even if the sorbent bed accidentally dries
- Broad selectivity: Retains compounds with log P values ranging from -2 to 10
During loading, pesticides are retained through hydrophobic interactions with the divinylbenzene components, while the hydrophilic N-vinylpyrrolidone groups help maintain sorbent wettability and reduce non-specific binding of polar matrix components.
Washing with Aqueous Solution to Remove Sugars and Organic Acids
After sample loading, a washing step is employed to remove water-soluble matrix components while retaining the target pesticides. Typically, 3-5 mL of 5-10% methanol in water is used as the wash solvent. This moderately polar solution effectively elutes sugars, organic acids, and other polar interferences while maintaining strong retention of most pesticide compounds.
Fruit matrices contain significant amounts of sugars (fructose, glucose, sucrose) and organic acids (citric, malic, tartaric acids) that can interfere with subsequent LC-MS/MS analysis. The wash step specifically targets these compounds:
- Sugars: Highly water-soluble compounds that elute readily in aqueous wash
- Organic acids: Polar acidic compounds that show minimal retention on HLB under neutral pH conditions
- Pigments: Some fruit pigments may be partially removed depending on their polarity
- Inorganic salts: Water-soluble salts from the QuEChERS extraction
The wash volume and composition can be optimized for specific fruit types. For example, citrus fruits with higher acid content may require slightly different wash conditions compared to sweeter fruits like grapes or apples.
Elution with Acetonitrile or Methanol
Pesticides are eluted from the HLB cartridge using a strong organic solvent, typically 3-5 mL of acetonitrile or methanol. Acetonitrile is often preferred for LC-MS/MS applications due to its lower viscosity and better compatibility with mass spectrometry detection. Methanol may be used when analyzing more polar pesticides that require stronger elution conditions.
The elution process typically involves:
- Initial elution: 2-3 mL of acetonitrile to displace water from the sorbent bed
- Secondary elution: Additional 1-2 mL to ensure complete recovery of retained pesticides
- Collection: Eluate is collected in a clean glass or polypropylene tube
For particularly hydrophobic pesticides or when analyzing complex fruit matrices, a small percentage of acid (0.1-1% formic acid) or base may be added to the elution solvent to disrupt any secondary interactions and improve recovery.
Evaporation and Reconstitution for LC-MS/MS Analysis
The eluate is concentrated by evaporation under a gentle stream of nitrogen at 30-40°C to near dryness. Care must be taken to avoid complete drying, as some pesticides may be lost through volatilization or adsorption to the tube walls. The evaporation step typically reduces the volume from 3-5 mL to 0.5-1 mL.
Reconstitution is performed using a solvent compatible with the LC-MS/MS mobile phase, typically:
- Initial mobile phase: Often water or aqueous buffer with 0.1% formic acid
- Organic modifier: May include 10-50% acetonitrile or methanol to ensure complete dissolution
- Volume adjustment: Final volume adjusted to 0.5-1.0 mL for optimal injection
The reconstituted sample may be filtered through a 0.2 μm syringe filter to remove any particulate matter before LC-MS/MS analysis. For maximum sensitivity, the reconstitution volume can be minimized to concentrate the analytes further.
Recovery Evaluation Across Multiple Pesticide Classes
Method validation requires comprehensive recovery studies across the target pesticide classes. Typical recovery acceptance criteria are 70-120% with RSDs <20% at regulatory limits. The HLB SPE method demonstrates excellent recovery for diverse pesticide classes:
Organophosphates and Carbamates
Moderately polar compounds with log P values typically ranging from 1-4 show excellent recovery (85-110%) on HLB cartridges. Their retention is primarily through hydrophobic interactions, with some hydrogen bonding possible through the N-vinylpyrrolidone groups.
Triazoles and Strobilurins
These fungicides, with log P values of 2-4, exhibit strong retention on HLB and typically achieve recoveries of 90-105%. Their aromatic structures facilitate π-π interactions with the divinylbenzene components.
Neonicotinoids
More polar insecticides with log P values around 0-2 require careful method optimization. Recovery can be improved by using weaker wash conditions and ensuring proper pH control during sample loading.
Pyrethroids
Highly hydrophobic compounds (log P 5-7) show excellent retention and recovery (95-110%) but may require stronger elution conditions or addition of small amounts of dichloromethane to the elution solvent.
Acidic Herbicides
For phenoxyacid herbicides and other acidic compounds, pH adjustment of the sample to suppress ionization is crucial for effective retention on HLB sorbents. Recovery typically ranges from 80-100% with proper pH control.
Studies have demonstrated that “SPE applications for food and beverages are usually developed either for quality control purposes or for the detection or identification of drug and pesticide residues or microbial toxins. The goal is to ensure the safety of the consumer.” The HLB SPE method, when properly optimized, provides reliable clean-up and concentration for multiresidue pesticide analysis in fruit samples, meeting the stringent requirements of modern food safety laboratories.
For laboratories seeking high-throughput solutions, 96-well SPE plates with HLB sorbent are available, enabling simultaneous processing of multiple samples. These plates maintain the same retention characteristics as cartridge formats while offering automation compatibility and improved workflow efficiency.
When selecting HLB SPE products for pesticide analysis in fruits, consider factors such as sorbent mass (typically 60-500 mg depending on sample load), particle size (30-60 μm for optimal flow characteristics), and format compatibility with your laboratory equipment. Proper method development and validation, including recovery studies with representative fruit matrices and pesticide spikes at relevant concentration levels, are essential for reliable results.