Common Pesticide Residues in Fruit Samples
Fruit samples present unique challenges for pesticide residue analysis due to their complex matrices containing sugars, organic acids, pigments, and varying water content. The most commonly encountered pesticide residues in fruits span multiple chemical classes, each requiring specific analytical approaches. Organochlorine pesticides such as DDT, DDE, DDD, endosulfan, and methoxychlor persist in the environment and can accumulate in fruit tissues. Organophosphates like chlorpyrifos, diazinon, and malathion represent another significant class, though they tend to be less persistent than organochlorines.
Carbamate insecticides, including carbaryl and methomyl, are frequently detected in fruit samples due to their widespread use in agriculture. Pyrethroids such as permethrin, cypermethrin, and deltamethrin have gained popularity as replacements for more toxic pesticides. Fungicides like benzimidazoles (thiabendazole, carbendazim) and triazoles (myclobutanil, tebuconazole) are particularly relevant for fruits susceptible to fungal diseases during storage and transport.
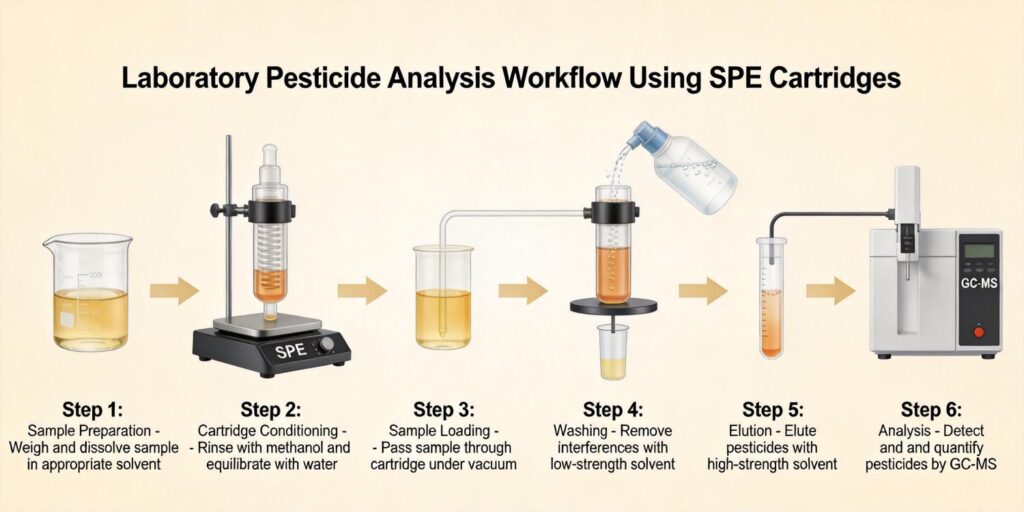
The Luke procedure, a comprehensive multiresidue method developed for pesticide analysis in fruits and vegetables, demonstrates the complexity of analyzing these matrices. This method has evolved from simple liquid-liquid extraction to incorporate multiple SPE steps for matrix removal and analyte concentration, highlighting the need for robust cleanup strategies in fruit analysis.
Homogenization and Extraction Solvent Selection
Proper sample preparation begins with homogenization, which ensures representative sampling and efficient extraction. For fruits, homogenization typically involves chopping or blending the entire fruit (including peel when appropriate) to create a uniform matrix. The high water content of most fruits (typically 80-90%) means that extraction often begins with water-miscible solvents.
Acetone is frequently employed as an initial extraction solvent for fruits due to its ability to penetrate plant tissues and extract a wide range of pesticides with varying polarities. Studies have demonstrated the effectiveness of acetone for extracting benzimidazole fungicides from various fruits and vegetables, followed by SPE cleanup on bonded sorbents. Acetonitrile has gained popularity in modern methods like QuEChERS (Quick, Easy, Cheap, Effective, Rugged, and Safe) for multiresidue pesticide analysis in fruits, offering excellent extraction efficiency while minimizing co-extraction of sugars and pigments.
The selection of extraction solvent must consider several factors: compatibility with subsequent SPE steps, ability to penetrate fruit tissues, and efficiency in extracting target pesticides while minimizing interference from matrix components. For fruits with high wax content (such as apples), post-extraction cleanup strategies may include selective precipitation of waxes using solvents like methanol, which dissolves pesticides while leaving waxes insoluble.
SPE Cleanup Using HLB Cartridges
Hydrophilic-Lipophilic Balanced (HLB) cartridges represent a versatile solution for pesticide cleanup in fruit extracts. These polymeric sorbents contain both hydrophilic N-vinylpyrrolidone and lipophilic divinylbenzene monomers, providing balanced retention for compounds across a wide polarity range. The HLB mechanism is particularly advantageous for fruit extracts because it effectively retains pesticides while allowing many polar matrix interferences (sugars, organic acids) to pass through.
For fruit sample cleanup, HLB cartridges are typically conditioned with methanol followed by water or a water-miscible solvent. The fruit extract, often diluted with water to reduce solvent strength, is then loaded onto the cartridge. The balanced nature of HLB sorbents ensures retention of both polar and non-polar pesticides, making them suitable for multiresidue methods targeting diverse chemical classes.
Research has demonstrated the effectiveness of HLB cartridges in various applications, including the analysis of fat-soluble vitamins in feed samples, where they provide excellent cleanup of complex matrices. For pesticide analysis in fruits, HLB cartridges offer several advantages: high capacity for both hydrophilic and hydrophobic compounds, stability across a wide pH range (pH 1-14), and compatibility with 100% aqueous conditions during loading.
Removal of Sugars and Pigments
Fruit matrices present significant challenges due to their high content of sugars (fructose, glucose, sucrose) and pigments (chlorophylls, carotenoids, anthocyanins). These compounds can interfere with chromatographic analysis, contaminate instrumentation, and mask target analytes. Effective removal of these interferences is crucial for accurate pesticide quantification.
Primary-Secondary Amine (PSA) sorbents are particularly effective for removing sugars and organic acids from fruit extracts. The amino groups on PSA interact with carbonyl groups of sugars through hydrogen bonding and weak anion exchange mechanisms. In the Luke procedure for multiresidue pesticide analysis, PSA sorbents are employed specifically to remove plant sugars and acids while allowing pesticides to pass through unretained.
For pigment removal, graphitized carbon black (GCB) or carbon-based sorbents offer excellent selectivity. These sorbents effectively trap planar molecules like chlorophylls and carotenoids through π-π interactions while allowing most pesticides to elute. However, care must be taken as some planar pesticides (such as certain pyrethroids) may also be retained on carbon sorbents. Mixed-mode sorbents combining carbon black with aminopropyl silica or PSA provide comprehensive cleanup for complex fruit matrices, as demonstrated in Japanese Ministry of Health methods for pesticide analysis in foods.
Anion exchange (SAX) cartridges can be employed to remove acidic pigments and organic acids, particularly when analyzing fruits with high anthocyanin content (berries, grapes). Stacked cartridge configurations, such as SAX/PSA combinations, provide sequential removal of different interference classes, significantly improving cleanup efficiency.
Elution Solvents Compatible with GC-MS
The final elution step in SPE cleanup must provide quantitative recovery of target pesticides while being compatible with subsequent GC-MS analysis. Solvent selection depends on the sorbent type, pesticide properties, and analytical requirements. For HLB cartridges used in fruit pesticide analysis, typical elution solvents include ethyl acetate, acetone, acetonitrile, or mixtures of these with non-polar solvents.
Ethyl acetate offers excellent elution strength for a wide range of pesticides while being sufficiently volatile for concentration steps prior to GC-MS analysis. Its moderate polarity makes it suitable for eluting both polar and non-polar compounds from HLB sorbents. Acetone provides stronger elution power and is particularly effective for more polar pesticides, though its higher boiling point requires longer evaporation times.
For GC-MS compatibility, solvent exchange may be necessary if the initial elution solvent is not ideal for injection. Common practice involves eluting with a solvent like ethyl acetate or acetone, then evaporating to near dryness and reconstituting in a solvent compatible with GC injection, typically hexane, iso-octane, or toluene. These solvents provide good solubility for most pesticides while minimizing peak broadening and decomposition in the GC inlet.
When analyzing pesticides requiring derivatization for GC analysis (such as chlorophenoxy acids), the elution solvent should be compatible with the derivatization reagents. Studies have shown that methylating chlorophenoxy acids before SPE cleanup allows them to pass through SAX/PSA combination cartridges without retention, demonstrating the importance of considering derivatization requirements in solvent selection.
Modern approaches often employ solvent mixtures optimized for specific pesticide classes. For example, hexane/acetone mixtures (90:10 v/v) have been successfully used for eluting organochlorine pesticides from Florisil cartridges in fruit analysis. The development of on-line SPE-GC systems has further refined solvent requirements, with systems demonstrating sub-ppt detection limits for pesticides in aqueous samples using optimized elution and transfer conditions.
Optimizing SPE Conditions for Fruit Analysis
Successful SPE cleanup for fruit pesticide analysis requires optimization of several parameters beyond solvent selection. Sample loading conditions must be adjusted based on fruit type and extraction method. Dilution of fruit extracts with water is often necessary to reduce solvent strength and ensure adequate retention on SPE cartridges. The high water content of fruit extracts (typically 70-90% water after homogenization) can be advantageous for reversed-phase SPE, as it promotes retention of hydrophobic pesticides.
pH adjustment may be necessary when analyzing ionizable pesticides. For acidic compounds (such as phenoxy acid herbicides), acidification of the sample ensures they remain in neutral form for retention on reversed-phase sorbents. Conversely, basic pesticides may require alkalization to prevent ionization and ensure proper retention.
Cartridge drying between wash and elution steps is critical for fruit extracts, as residual water can interfere with elution efficiency and subsequent solvent evaporation. Vacuum drying for 15-30 minutes is typically employed, though care must be taken to avoid excessive drying that could lead to pesticide degradation or irreversible binding to the sorbent.
Quality Control Considerations
Implementing appropriate quality control measures is essential for reliable pesticide analysis in fruits. Matrix-matched calibration standards should be prepared using pesticide-free fruit extracts to account for matrix effects. Recovery studies using fortified samples at multiple concentration levels provide validation of the entire analytical procedure, from extraction through SPE cleanup to final analysis.
Procedural blanks should be included to monitor for contamination from SPE cartridges, solvents, or laboratory environment. The low extractable levels of modern SPE cartridges (as low as 5 ppm total extractables for high-quality products) minimize background interference in sensitive GC-MS analyses.
Method validation should include assessment of precision, accuracy, limits of detection and quantification, and robustness against variations in fruit type and maturity. The comprehensive nature of fruit matrices necessitates thorough validation to ensure method reliability across different fruit commodities.
Conclusion
Solid phase extraction remains an indispensable tool for pesticide residue analysis in fruits, providing the necessary cleanup to overcome complex matrix interferences. The combination of HLB cartridges for broad-spectrum pesticide retention with specialized sorbents for sugar and pigment removal creates a powerful cleanup strategy. Proper solvent selection and optimization of SPE conditions ensure compatibility with sensitive GC-MS detection, enabling accurate quantification of pesticides at trace levels. As regulatory requirements become more stringent and the number of target pesticides expands, robust SPE methods will continue to play a critical role in ensuring food safety and compliance with maximum residue limits.