Honey Matrix Challenges Including High Sugar Content
Honey presents unique analytical challenges for veterinary antibiotic residue analysis due to its complex matrix composition. As a natural product with approximately 80% sugar content (primarily fructose and glucose), honey creates significant interference issues for analytical methods. The high viscosity and sugar concentration can lead to:
- Column fouling and reduced chromatographic performance
- Matrix effects in mass spectrometry detection
- Reduced extraction efficiency for target analytes
- Increased background noise and reduced signal-to-noise ratios
According to veterinary drug abuse literature, complex matrices like honey require specialized extraction approaches to overcome these challenges. The high sugar content can compete with target analytes for binding sites on SPE sorbents and create viscous solutions that impede proper sample flow through extraction cartridges.
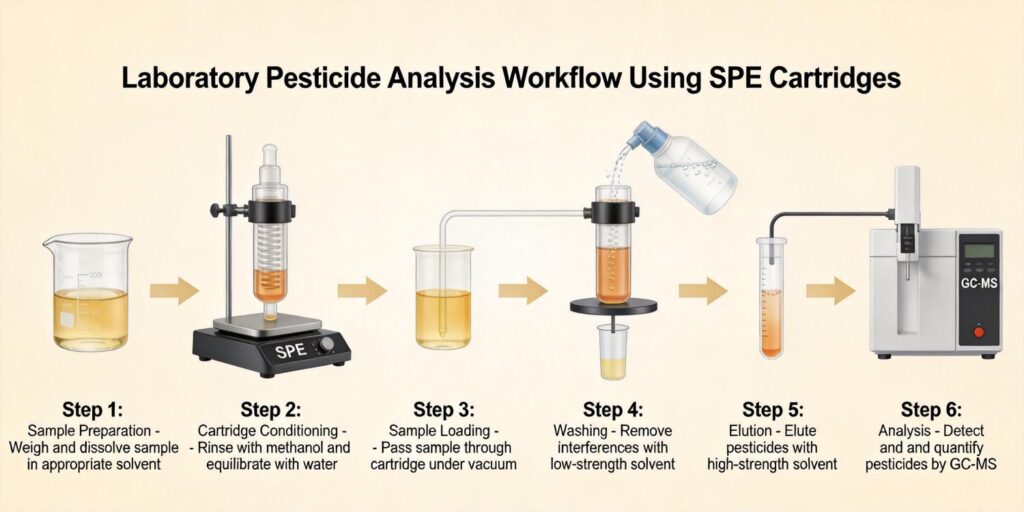
Dilution of Honey with Water Followed by Filtration
The initial sample preparation step is critical for successful antibiotic extraction from honey. A standardized approach involves:
- Sample Homogenization: Thoroughly mix the honey sample to ensure uniform distribution of potential antibiotic residues.
- Dilution Protocol: Typically, a 1:1 to 1:5 (w/v) dilution with purified water or buffer solution is employed. This reduces viscosity and sugar concentration while maintaining analyte solubility.
- pH Adjustment: Depending on the target antibiotics, pH may be adjusted to optimize extraction efficiency. For broad-spectrum antibiotic screening, neutral pH (6.0-7.0) is often recommended.
- Filtration: Pass the diluted honey solution through a 0.45 μm membrane filter to remove particulate matter that could clog SPE cartridges.
Research indicates that proper dilution is essential for preventing cartridge clogging and ensuring consistent flow rates during SPE processing. The dilution factor should be optimized to balance matrix effects with analyte concentration requirements for detection.
Selection of HLB SPE Cartridge for Antibiotic Retention
The Hydrophilic-Lipophilic Balance (HLB) SPE cartridge represents the optimal choice for veterinary antibiotic extraction from honey for several technical reasons:
HLB Sorbent Chemistry
HLB cartridges contain a unique copolymer of divinylbenzene and N-vinylpyrrolidone that provides:
- Dual Retention Mechanism: Both hydrophilic and lipophilic interactions for broad-spectrum antibiotic coverage
- High Capacity: Typically 15-30 mg per 1 cc cartridge for efficient antibiotic retention
- pH Stability: Effective performance across pH 1-14, crucial for antibiotics with varying pKa values
- Wetting Characteristics: Does not require complete drying between steps, reducing analyte loss
Antibiotic Compatibility
HLB sorbents effectively retain various antibiotic classes commonly used in veterinary medicine:
- Tetracyclines: Oxytetracycline, chlortetracycline, doxycycline
- Sulfonamides: Sulfadimethoxine, sulfamethazine, sulfathiazole
- Macrolides: Erythromycin, tylosin, tilmicosin
- Fluoroquinolones: Enrofloxacin, ciprofloxacin, danofloxacin
- Aminoglycosides: Streptomycin, gentamicin, neomycin
Studies have demonstrated that mixed-mode sorbents like HLB provide cleaner extracts than traditional C18 phases when dealing with complex matrices like honey, as noted in veterinary drug analysis literature.
Cartridge Conditioning Protocol
Proper conditioning of HLB cartridges is essential for optimal antibiotic recovery from honey samples. The recommended protocol includes:
- Methanol Activation: Pass 1-2 mL of HPLC-grade methanol through the cartridge at approximately 1 mL/min flow rate. This step solvates the polymer and opens the pore structure.
- Water Equilibration: Follow with 1-2 mL of purified water or appropriate buffer solution to remove methanol and prepare the sorbent for aqueous sample loading.
- Conditioning Solution Compatibility: Ensure the final conditioning solution matches the sample matrix pH and ionic strength to prevent premature analyte elution.
- Flow Rate Control: Maintain consistent flow rates (typically 1-3 mL/min) throughout conditioning to ensure uniform sorbent preparation.
Research indicates that incomplete conditioning can lead to reduced recovery rates, particularly for polar antibiotics that require proper sorbent hydration for effective retention.
Loading Diluted Honey Extract
The sample loading process requires careful attention to several parameters:
Loading Conditions
- Flow Rate: 1-2 mL/min to ensure adequate contact time between analytes and sorbent
- Sample Volume: Typically 1-10 mL depending on antibiotic concentration and detection limits
- pH Control: Maintain pH within optimal range for target antibiotic classes (often pH 6-7 for broad screening)
- Temperature: Room temperature (20-25°C) to maintain consistent extraction characteristics
Loading Solution Optimization
Based on fat-soluble vitamin extraction studies using HLB cartridges, the ethanol concentration in the loading solution significantly affects retention. For honey antibiotic extraction:
- Maintain organic solvent concentration below 5% to ensure antibiotic retention
- Consider adding appropriate buffers to maintain ionic strength and pH
- Monitor solution clarity to prevent particulate matter from entering the cartridge
Washing with Water to Remove Sugars
The washing step is critical for removing interfering sugars and other honey matrix components while retaining target antibiotics:
- Primary Wash: 1-2 mL of purified water to remove residual sugars and polar interferences
- Secondary Wash: Optional wash with 5% methanol in water to remove moderately polar interferences without eluting antibiotics
- Cartridge Drying: Apply vacuum or positive pressure (5-10 psi) for 1-2 minutes to remove residual water before elution
- Wash Volume Optimization: Determine optimal wash volume through method development to balance cleanliness and recovery
Studies on veterinary drug extraction emphasize the importance of selective washing to remove endogenous compounds while maintaining high antibiotic recovery rates. The high sugar content of honey makes this step particularly crucial for obtaining clean extracts suitable for sensitive detection methods.
Elution with Methanol Containing Acid Modifier
The elution step requires careful solvent selection to efficiently recover retained antibiotics:
Elution Solvent Composition
- Primary Solvent: HPLC-grade methanol provides excellent elution strength for most antibiotics
- Acid Modifier: 0.1-2% formic acid or acetic acid to protonate basic antibiotics and improve recovery
- Alternative Modifiers: For certain antibiotic classes, ammonium hydroxide or other basic modifiers may be appropriate
- Solvent Volume: Typically 1-3 mL, collected in two fractions to ensure complete elution
Elution Protocol
- Apply elution solvent slowly (0.5-1 mL/min) to maximize contact time
- Collect eluate in clean glass or polypropylene tubes
- Evaporate to dryness under gentle nitrogen stream at 40-50°C
- Reconstitute in mobile phase compatible solvent for analysis
Research indicates that acid-modified methanol provides superior recovery for many veterinary antibiotics compared to pure organic solvents, particularly for compounds with basic functional groups.
LC-MS/MS Analysis of Antibiotic Residues
The final analytical step employs liquid chromatography-tandem mass spectrometry for sensitive and specific antibiotic detection:
Chromatographic Conditions
- Column Selection: C18 or equivalent reversed-phase column (e.g., 100 × 2.1 mm, 1.7-3 μm particle size)
- Mobile Phase: Gradient elution with water and methanol/acetonitrile containing 0.1% formic acid
- Flow Rate: 0.2-0.4 mL/min for optimal separation and MS compatibility
- Column Temperature: 30-40°C for consistent retention times
Mass Spectrometry Parameters
- Ionization Mode: Electrospray ionization (ESI) in positive or negative mode depending on antibiotic class
- Multiple Reaction Monitoring (MRM): Two transitions per antibiotic for confirmation
- Collision Energy: Optimized for each target compound
- Source Parameters: Optimized desolvation temperature, gas flows, and capillary voltage
Method Validation
According to veterinary drug analysis standards, the complete method should be validated for:
- Linearity: Correlation coefficient (r²) > 0.99 over relevant concentration range
- Recovery: 70-120% for most antibiotics at relevant concentrations
- Precision: Relative standard deviation < 15% for repeatability and reproducibility
- Limit of Detection/Quantification: Appropriate for regulatory requirements (typically 1-10 μg/kg)
- Specificity: No interference from honey matrix components at retention times of target antibiotics
The combination of HLB SPE extraction with LC-MS/MS analysis provides a robust, sensitive method for veterinary antibiotic residue analysis in honey. This approach addresses the unique challenges of the honey matrix while delivering the specificity and sensitivity required for regulatory compliance and food safety monitoring.
For laboratories requiring high-throughput analysis, this method can be adapted to 96-well SPE plate formats while maintaining the extraction efficiency of individual HLB cartridges. The versatility of HLB chemistry makes it suitable for various antibiotic classes, providing laboratories with a single extraction method for comprehensive antibiotic screening in honey products.