Antibiotic Residue Regulations Overview
Antibiotic residue analysis in milk represents a critical component of food safety monitoring worldwide. Regulatory frameworks such as the European Union’s Maximum Residue Limits (MRLs), the U.S. Food and Drug Administration’s tolerances, and China’s GB standards establish stringent requirements for veterinary drug residues in dairy products. These regulations typically specify detection limits in the parts-per-billion (ppb) range, necessitating highly sensitive analytical methods.
According to research documented in the literature, milk presents unique analytical challenges due to its complex matrix composition. As noted in the Solid Phase Extraction: Principles, Techniques and Applications reference, “Milk, butter and cheese present a bigger problem. They have a high fat and protein content and they are quite viscous or completely solid.” This complexity requires specialized sample preparation approaches to achieve the necessary sensitivity and selectivity for regulatory compliance.
Sample Pretreatment for Milk Matrices
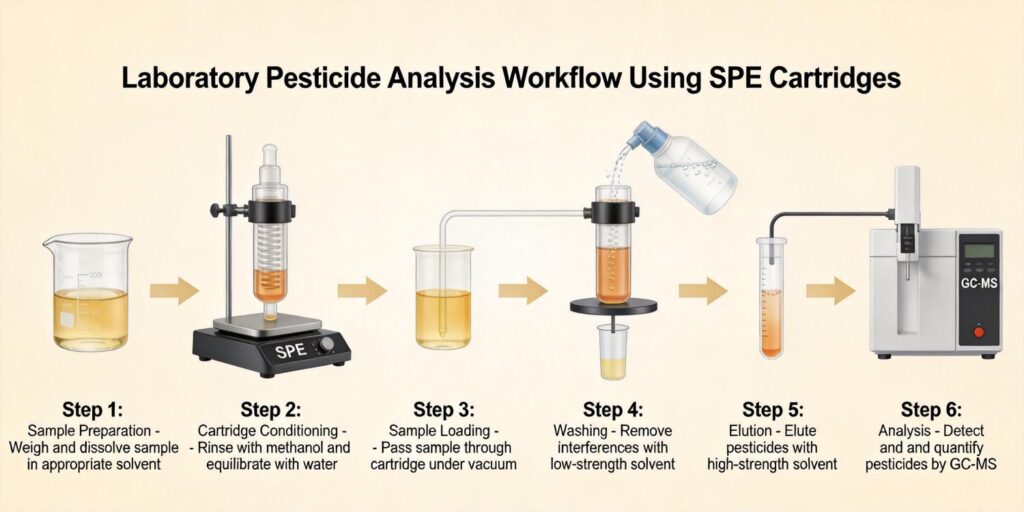
Proper sample pretreatment is essential for successful antibiotic residue analysis in milk. The initial step typically involves homogenization to ensure representative sampling, followed by appropriate dilution to reduce matrix effects. For HLB (Hydrophilic-Lipophilic Balance) SPE applications, sample pretreatment often includes acidification or buffering to optimize analyte retention characteristics.
Research by Long et al. (1990) demonstrated effective approaches for antibiotic residue extraction from milk matrices, establishing foundational methodologies that continue to inform contemporary analytical practices. The pretreatment step must balance analyte recovery with effective removal of interfering matrix components.
Protein Precipitation Prior to SPE
Protein precipitation represents a critical preliminary step in milk sample preparation for antibiotic analysis. Common approaches include:
- Acetonitrile precipitation (typically 1:2 or 1:3 sample:acetonitrile ratio)
- Trichloroacetic acid precipitation
- Acidified methanol precipitation
As documented in Waters Oasis documentation, “Protein precipitation (PPT) 1:3 Plasma:ACN” represents a common approach, though SPE methods often provide superior cleanup. The precipitation step serves to denature and remove milk proteins that could otherwise interfere with SPE cartridge performance and subsequent LC-MS/MS analysis.
HLB Cartridge Conditioning Protocol
Proper conditioning of HLB cartridges is essential for optimal performance in antibiotic residue analysis. The standard conditioning protocol for Poseidon Scientific HLB cartridges includes:
- Methanol activation: 3-5 mL methanol to wet the polymeric sorbent
- Water equilibration: 3-5 mL deionized water to create aqueous-compatible conditions
It’s important to note that some advanced HLB formulations, such as Oasis PRiME HLB, eliminate the need for conditioning steps, as noted in product documentation: “Directly load pre-treated samples without conditioning and equilibration.” However, for traditional HLB cartridges, proper conditioning remains essential for reproducible analyte recovery.
Loading Diluted Milk Extract
Following protein precipitation and centrifugation, the supernatant requires appropriate dilution before loading onto the HLB cartridge. Typical dilution approaches include:
- 1:1 dilution with acidified water (0.1-1% formic acid)
- Dilution with buffer solutions to optimize pH for specific antibiotic classes
- Maintaining organic solvent content below 10% to ensure proper analyte retention
Loading rates should be controlled to 1-2 mL/min to ensure adequate contact time between analytes and the HLB sorbent. Research indicates that flow rate optimization significantly impacts recovery rates for antibiotic compounds in milk matrices.
Wash Solvent to Remove Fats and Proteins
Effective washing represents a critical step in removing residual fats, proteins, and other matrix interferences while retaining target antibiotic analytes. Common wash protocols for milk antibiotic analysis include:
- 5% methanol in water: Effective for removing polar interferences while retaining most antibiotics
- Water washes: Additional aqueous washes to remove salts and sugars
- Low-percentage organic washes: Typically 5-10% methanol or acetonitrile in water
As documented in SPE literature, “Wash: 5% MeOH” represents a standard approach for HLB cartridges in biological sample preparation. The wash step must be carefully optimized to balance interference removal with analyte retention.
Elution Using Methanol/Acetonitrile Mixture
Elution represents the final step in the SPE process, where target antibiotics are recovered from the HLB cartridge. For antibiotic residue analysis in milk, optimal elution typically involves:
- Methanol/acetonitrile mixtures: Commonly 90:10 or 80:20 acetonitrile:methanol ratios
- Acidified or basified eluents: Depending on antibiotic pKa values
- Volume optimization: Typically 2-5 mL elution volume for adequate recovery
Documentation from Waters specifies “Elute: 90/10 acetonitrile/MeOH” as an effective elution protocol for HLB cartridges. The elution solvent must effectively disrupt analyte-sorbent interactions while maintaining compatibility with subsequent LC-MS/MS analysis.
LC-MS/MS Analysis Workflow
Following SPE cleanup, the eluate undergoes concentration (typically nitrogen evaporation) and reconstitution in mobile phase-compatible solvents before LC-MS/MS analysis. Key considerations include:
Chromatographic Conditions
Reverse-phase chromatography using C18 or C8 columns represents the standard approach for antibiotic separation. Mobile phase gradients typically employ water and methanol or acetonitrile with acid modifiers (0.1% formic acid being common).
Mass Spectrometric Detection
Triple quadrupole MS/MS systems operating in multiple reaction monitoring (MRM) mode provide the sensitivity and selectivity required for antibiotic residue analysis at regulatory limits. Key parameters include:
- Electrospray ionization (ESI) in positive or negative mode depending on analyte properties
- Optimized collision energies for each antibiotic compound
- Appropriate internal standards for quantification
Method Validation
Validated methods must demonstrate adequate sensitivity (typically ≤ 1 ppb), precision (RSD < 15-20%), accuracy (80-120% recovery), and linearity across the required concentration range. Matrix-matched calibration standards are essential for accurate quantification.
Conclusion
HLB SPE represents a robust and versatile approach for antibiotic residue analysis in milk matrices. The hydrophilic-lipophilic balanced chemistry of HLB sorbents provides effective retention for a wide range of antibiotic classes while enabling efficient removal of milk matrix interferences. When combined with optimized sample pretreatment and LC-MS/MS detection, HLB SPE methods deliver the sensitivity, selectivity, and reproducibility required for regulatory compliance in dairy product safety monitoring.
For laboratories seeking reliable SPE solutions for antibiotic analysis, Poseidon Scientific’s HLB cartridges offer consistent performance and compatibility with established analytical protocols. The method outlined here provides a foundation that can be further optimized for specific antibiotic classes or regulatory requirements.