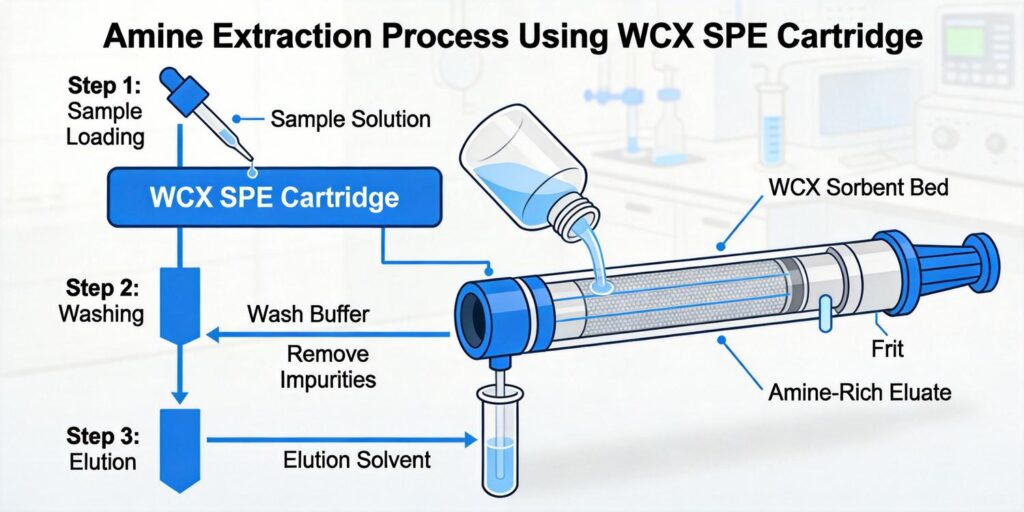
Why WCX Works Well for Peptides: Understanding pKa Behavior
Weak Cation Exchange (WCX) solid-phase extraction represents a sophisticated mixed-mode approach that combines both ion-exchange and reversed-phase retention mechanisms. This dual functionality makes WCX particularly effective for peptide sample cleanup, especially when dealing with strong bases and quaternary amines. The key to understanding WCX’s effectiveness lies in the pKa behavior of peptides and the sorbent’s chemical properties.
Peptides typically contain multiple ionizable groups, including amino termini (pKa ~8-10), carboxyl termini (pKa ~2-4), and various side chains. The WCX sorbent features weak cation exchange groups that interact with positively charged peptide molecules. According to Waters documentation, WCX is specifically designed for strong bases with pKa >10, making it ideal for peptides that maintain positive charges across a wide pH range. The mixed-mode retention ensures that even when ionic interactions are disrupted, hydrophobic interactions continue to retain peptides, providing a safety net against sample loss.
The polymeric nature of modern WCX sorbents, such as those from Poseidon Scientific, offers pH stability from 0-14, allowing for flexible method development. This wide pH compatibility is crucial when working with peptides that may require extreme pH conditions for optimal extraction. The water-wettable properties of these sorbents ensure consistent performance even with highly aqueous samples, a common scenario in proteomics workflows.
Selecting Loading pH Near Peptide Isoelectric Point
The isoelectric point (pI) of a peptide represents the pH at which it carries no net electrical charge. For effective WCX extraction, loading samples at a pH near or slightly below the peptide’s pI is critical. When the pH is below the pI, peptides carry a net positive charge, enabling strong ionic interactions with the negatively charged WCX functional groups.
To determine the optimal loading pH:
- Calculate or experimentally determine the peptide’s pI using available software or titration methods
- Set loading pH 1-2 units below the pI to ensure protonation of basic groups
- Consider using ammonium acetate or phosphate buffers in the range of pH 4-6 for most peptides
- For basic peptides with high pI values (>9), loading at pH 7-8 may be appropriate
Research indicates that maintaining the loading pH within this optimal range maximizes ionic interactions while minimizing non-specific binding. The mixed-mode nature of WCX ensures that even if ionic interactions are suboptimal, hydrophobic interactions will still provide adequate retention.
Cartridge Conditioning Procedure
Proper conditioning of WCX cartridges is essential for reproducible peptide recovery. The conditioning process prepares the sorbent for optimal interaction with your samples. Follow this standardized procedure:
- Solvation: Apply 1-2 column volumes of methanol or acetonitrile to wet the hydrophobic polymer backbone. For a 30 mg cartridge, use approximately 1 mL of organic solvent.
- Equilibration: Follow with 1-2 column volumes of deionized water to remove excess organic solvent that could interfere with ionic interactions.
- pH Adjustment: Apply 1-2 column volumes of loading buffer (typically the same buffer used for sample dilution) to establish the correct pH environment for ionic exchange.
Critical considerations during conditioning include maintaining a consistent flow rate (1-2 mL/min for manual processing) and preventing the sorbent from drying between steps. If the sorbent accidentally dries, repeat the conditioning process from the beginning. For automated systems, ensure proper vacuum settings (typically 2-3 inches Hg) to maintain optimal sorbent hydration.
Sample Dilution Buffers for Peptide Stability
Choosing appropriate dilution buffers is crucial for maintaining peptide stability during the extraction process. The buffer must serve multiple functions: maintaining optimal pH for ionic interactions, preventing peptide degradation, and minimizing non-specific binding.
Recommended buffer systems include:
- Ammonium acetate: 10-50 mM, pH 4-6, volatile for MS compatibility
- Phosphate buffers: 10-50 mM, pH 4-6, excellent buffering capacity
- Formic acid: 0.1-1% in water, pH ~2.5, for highly basic peptides
For plasma or serum samples, Waters documentation recommends diluting with 4% phosphoric acid before loading onto WCX cartridges. This acidification helps precipitate proteins while maintaining peptides in solution. The typical dilution ratio is 1:3 to 1:4 (sample:buffer), depending on sample complexity and peptide concentration.
Add protease inhibitors when working with biological samples to prevent peptide degradation during processing. Maintain samples at 4°C during the extraction process to further enhance stability.
Salt Removal Using Aqueous Wash
Effective salt removal is essential for downstream analytical techniques, particularly mass spectrometry. The aqueous wash step in WCX protocols serves to remove salts, polar contaminants, and residual matrix components while retaining peptides through ionic interactions.
Standard aqueous wash conditions include:
- 2-5% organic solvent (methanol or acetonitrile) in water or buffer
- pH adjusted to maintain peptide ionization state
- 1-3 column volumes depending on sample complexity
For plasma samples, the first wash must be aqueous to remove plasma proteins. Waters protocols typically use 5% ammonium hydroxide as the first wash for peptide applications. This alkaline wash helps remove acidic contaminants while maintaining basic peptides in their protonated, positively charged state.
The flow rate during washing should be moderate (1-2 mL/min) to ensure thorough contaminant removal without prematurely eluting peptides. Monitor wash fractions if necessary to confirm that peptides are not being lost during this step.
Organic Wash for Hydrophobic Contaminants
Following aqueous washing, an organic wash step removes hydrophobic contaminants that could interfere with analysis. This step leverages the reversed-phase component of WCX’s mixed-mode mechanism.
Recommended organic wash conditions:
- 20-50% acetonitrile or methanol in water
- May include 0.1-1% formic acid or ammonium hydroxide for pH control
- 1-2 column volumes sufficient for most applications
Waters therapeutic peptide protocols typically use 20% acetonitrile as the second wash. This intermediate organic strength removes lipids, non-polar contaminants, and weakly retained compounds while maintaining peptide retention through ionic interactions.
The organic wash serves an additional purpose: it helps dry the sorbent bed in preparation for elution. For applications requiring complete drying before elution with water-immiscible solvents, extend the organic wash or include a dedicated drying step under vacuum.
Elution with Mild Acidic Organic Solvents
Peptide elution from WCX cartridges requires disruption of both ionic and hydrophobic interactions. Mild acidic organic solvents effectively achieve this while maintaining peptide integrity.
Optimal elution conditions:
- Solvent composition: 50-75% acetonitrile or methanol in water
- Acidification: 0.1-2% formic acid or trifluoroacetic acid (TFA)
- Volume: 1-3 column volumes, collected in minimal fractions
Waters protocols for peptide elution typically use 1% TFA in 75/25 acetonitrile/water. The acidic conditions (pH ~2) neutralize the positive charges on peptides, disrupting ionic interactions with the WCX sorbent. The high organic content simultaneously disrupts hydrophobic interactions.
For maximum recovery, allow the elution solvent to soak the sorbent for 30-60 seconds before applying vacuum or pressure. Collect eluate in small fractions (0.5-1 column volumes) to monitor elution profile and maximize concentration. The low elution volumes possible with modern SPE formats (as low as 25 μL with μElution plates) enable significant sample concentration without evaporation.
Recovery Testing with LC-MS Peptide Standards
Validating WCX peptide cleanup protocols requires systematic recovery testing using appropriate standards. This process ensures method robustness and identifies potential optimization needs.
Recommended approach for recovery testing:
- Select representative peptides: Choose standards covering your expected pI range, molecular weights, and hydrophobicities
- Spike into appropriate matrix: Use buffer, plasma, or other relevant matrices at concentrations relevant to your application
- Process through complete protocol: Include all steps from conditioning through elution
- Analyze by LC-MS: Compare peak areas or heights of extracted samples versus direct injections
- Calculate recovery: Recovery (%) = (Area extracted / Area direct injection) × 100
Waters therapeutic peptide method development kits include screening protocols that achieve recoveries >80% for most peptides. These kits utilize μElution plates that enable 15× sample concentration without evaporation, significantly enhancing sensitivity for low-abundance peptides.
When troubleshooting recovery issues, consider adjusting loading pH, wash stringency, or elution solvent composition. The mixed-mode nature of WCX provides multiple parameters for optimization to achieve optimal recovery for specific peptide classes.
Application in Proteomics Workflows
WCX SPE plays a crucial role in modern proteomics workflows, particularly in sample preparation for mass spectrometry analysis. The technique offers several advantages for proteomic applications:
- Desalting and buffer exchange: Removes salts and detergents incompatible with MS analysis
- Concentration: Enables analysis of low-abundance peptides from large sample volumes
- Selective enrichment: Can be tuned to enrich specific peptide classes based on charge properties
- Matrix cleanup: Removes contaminants that cause ion suppression in MS
In bottom-up proteomics, WCX is particularly valuable after tryptic digestion. Tryptic peptides typically contain C-terminal lysine or arginine residues, resulting in basic peptides well-suited for WCX retention. The technique can be combined with other enrichment strategies, such as TiO2 for phosphopeptides or lectin columns for glycopeptides, in multidimensional purification schemes.
For clinical proteomics, WCX enables cleanup of plasma or serum samples before MS analysis. The mixed-mode retention provides superior removal of abundant proteins and lipids compared to reversed-phase-only methods. This results in cleaner spectra and improved detection of low-abundance biomarkers.
The compatibility of WCX with automation makes it ideal for high-throughput proteomics. Ninety-six-well plate formats enable parallel processing of multiple samples, while μElution plates minimize final volumes for direct MS analysis. These features position WCX as a cornerstone technology in modern proteomics sample preparation pipelines.
Optimization Tips for Specific Applications
For researchers developing custom WCX protocols, consider these optimization strategies:
- For phosphopeptide analysis, include TiO2 enrichment after WCX cleanup
- For membrane proteomics, incorporate detergent removal steps before WCX
- For quantitative proteomics, ensure consistent recovery across all samples by standardizing loading amounts and wash conditions
- For biomarker discovery, use WCX in combination with depletion columns to remove high-abundance proteins
Poseidon Scientific’s WCX products offer the performance and consistency required for demanding proteomics applications. With proper method development and optimization, WCX SPE provides reliable, high-recovery peptide cleanup that enhances downstream analytical results.