Overview of Mixed-Mode Cation Exchange SPE Mechanisms
Mixed-mode solid phase extraction represents a sophisticated approach to sample preparation that combines multiple retention mechanisms within a single sorbent. For basic drug analysis, mixed-mode cation exchange (MCX and WCX) cartridges provide superior selectivity and sensitivity compared to traditional reversed-phase materials. These sorbents operate through a dual mechanism: reversed-phase hydrophobic interactions and ionic exchange, creating a powerful combination for isolating protonated amines from complex biological matrices.
According to forensic and clinical applications literature, mixed-mode SPE columns are prevalent in drug extractions because they offer multiple binding mechanisms for improved sensitivity and excellent sample cleanup. A mixed-mode sorbent employs two or more binding mechanisms in the same column, with reversed phase combined with ion exchangers being most common in drug extractions. When using mixed-mode extractions, the elution solvent must be able to reverse or disrupt all bonding mechanisms simultaneously, so pH, polarity, and solubility must all be considered.
The versatility of SPE can be best exhibited by its usage in the separation of a wide variety of drugs using a combination of separation strategies. The widest variety of separations have been developed using the combination of a C8 with a cationic exchanger, usually benzene sulfonic acid. This mechanism works by providing separation of the acid/neutral drugs using reversed phase C8 functionality, while the benzene sulfonic acid mechanism works on basic drugs by cation exchange of the amine functionalities.
Chemical Structure Differences Between MCX and WCX
MCX: Strong Cation Exchange with Sulfonic Acid Groups
MCX (Mixed-mode Cation eXchange) sorbents feature strong sulfonic acid groups (-SO₃H) as their ionic exchange sites. These sulfonic acid groups are permanently charged across the entire pH range (0-14), making them strong cation exchangers. The Oasis MCX sorbent has a tightly controlled ion-exchange capacity of 1 meq/g, ensuring reproducible SPE protocols for extraction of basic compounds and metabolites from biological fluids. There are no silanol groups to complicate the retention mode or method development.
WCX: Weak Cation Exchange with Carboxylic Acid Groups
WCX (Weak Cation eXchange) sorbents utilize weak carboxylic acid groups (-COOH) as their ionic exchange sites. These carboxylic acid groups are pH-dependent, with optimal performance in the pH range of 4-8. The Oasis WCX sorbent has a tightly controlled ion-exchange capacity of 0.7 meq/g and was specifically developed to provide better sample preparation for strong bases and quaternary amines. The retention mechanism is mixed mode (both ion-exchange and reversed-phase), which improves retention for all types of basic analytes, especially strong bases.
pH Behavior: MCX Stability vs WCX Optimal Range
The fundamental difference in pH behavior between MCX and WCX sorbents stems from their chemical structures. MCX sorbents, with their strong sulfonic acid groups, maintain consistent ionic exchange capacity across the entire pH range from 0-14. This pH stability makes method development simple and fast, with methods that are more robust and reliable.
In contrast, WCX sorbents with weak carboxylic acid groups exhibit pH-dependent behavior. The carboxylic acid groups are protonated (uncharged) at low pH and deprotonated (negatively charged) at higher pH. For optimal cation exchange performance, the pH should be maintained above the pKa of the carboxylic acid groups (typically pH 4-8). This pH sensitivity requires more careful control during method development but offers advantages for specific applications.
Retention Mechanisms for Protonated Amines
Both MCX and WCX sorbents retain protonated amines through a combination of mechanisms. The primary retention occurs through ionic interactions between the positively charged amine groups and the negatively charged exchange sites on the sorbent. Secondary retention occurs through reversed-phase hydrophobic interactions with the polymer backbone.
Research has shown that retention of organic bases using strong cation exchange sorbents like MCX is dependent on the protonation of the bases to their cationic form, and this mechanism is therefore capable of extracting compounds with a greater range of pKa values. This has particular application in the case of ring-hydroxylated metabolites whose high basicity, combined with high polarity, might result in poorer recovery by solvent extraction.
For weakly basic drugs such as benzocaine and diazepam, while the compounds initially retain well on the sorbent by non-polar interactions, their acid-base properties result in poor retention at the sulfonate sites and their subsequent elution in the methanolic wash. The 4-amino group of benzocaine is only weakly basic due to the destabilization of the protonated form by the electron withdrawing ester group.
Method Comparison Workflow
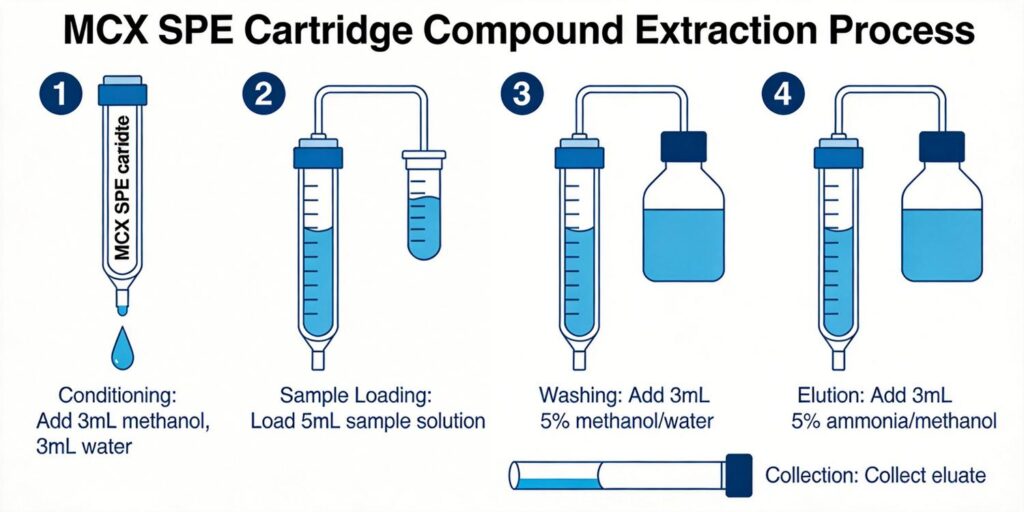
Conditioning Solvents
Both MCX and WCX cartridges require proper conditioning before use. Standard conditioning protocols involve rinsing with methanol followed by water or buffer. For cation exchange sorbents, conditioning typically includes 6 mL of methanol followed by 3 mL of buffer solution at appropriate pH (pH 4.5 for SCX sorbents).
Sample pH Adjustment
Sample pH adjustment is critical for optimal retention of basic compounds. For MCX sorbents, samples should be acidified to pH 2-3 to ensure protonation of basic compounds. For WCX sorbents, the optimal pH range is 4-8, where carboxylic acid groups are deprotonated and available for ionic exchange.
Wash Solvent Composition
Wash steps typically involve water or aqueous solutions to remove polar interferences. For mixed-mode cartridges, additional washes with organic solvents or acidic/basic solutions may be employed to remove specific interferences while retaining the analytes of interest.
Case Study: Extracting β-Blockers from Plasma
A comparative study of β-blocker extraction from plasma demonstrates the practical differences between MCX and WCX sorbents. Propranolol and atenolol, representing hydrophobic and hydrophilic β-blockers respectively, were studied using mixed-mode extraction.
Research has shown that the elution characteristics of β-blockers like propranolol and atenolol from mixed-mode cartridges differ significantly. Following application in water, cartridges were successively washed with portions of aqueous methanol where the concentration of methanol was increased from 0 to 100% in 20% increments. The cumulative elution over the whole series of solvents was never more than 7%, demonstrating strong retention.
MCX sorbents provided excellent recovery for both hydrophobic and hydrophilic β-blockers across a wide pH range. WCX sorbents showed optimal performance for strong bases like propranolol but required careful pH control for compounds like atenolol with different pKa values.
Elution Strategies
Basic Eluents for MCX
For MCX sorbents, elution typically employs basic solvents such as 5% NH₄OH in methanol. The ammonia neutralizes the sulfonic acid groups and disrupts the ionic interactions, while methanol provides the organic solvent strength needed for reversed-phase elution.
Neutral Organic Solvents for WCX
WCX sorbents can be eluted with neutral organic solvents or solvents with controlled pH. Since the carboxylic acid groups are pH-dependent, adjusting the elution solvent pH below 4 can protonate the exchange sites and release the retained bases.
Decision Matrix for Analysts
When to Choose MCX
- Analyzing basic compounds with pKa values < 10
- Working with samples requiring pH extremes (0-14)
- Need for maximum method robustness and reproducibility
- Extracting a wide range of basic compounds with varying properties
- Applications requiring high ion-exchange capacity (1 meq/g)
When to Choose WCX
- Analyzing strong bases and quaternary amines
- Samples with pH naturally in the 4-8 range
- Applications requiring selective retention of strong bases
- When gentler elution conditions are preferred
- Extracting compounds that may degrade under strongly basic conditions
General Recommendations
For method development, consider using Oasis Sorbent Selection Kits that include both MCX and WCX sorbents. These kits enable rapid development of SPE methods for LC-MS analysis by having all four Oasis Ion-exchange Sorbents (MCX, MAX, WAX, and WCX) in a single plate or cartridge kit, which is convenient for scouting the best methods to accomplish efficient isolation of unknown analytes, zwitterionic compounds, or mixtures of analytes with different retention/elution properties.
Ultimately, the choice between MCX and WCX depends on the specific analytes, sample matrix, and analytical requirements. MCX offers broader pH stability and higher capacity, making it suitable for general basic drug analysis. WCX provides specialized performance for strong bases and applications where pH control is manageable. Both sorbents represent significant advancements over traditional reversed-phase materials for basic drug extraction, offering improved selectivity, sensitivity, and sample cleanup.