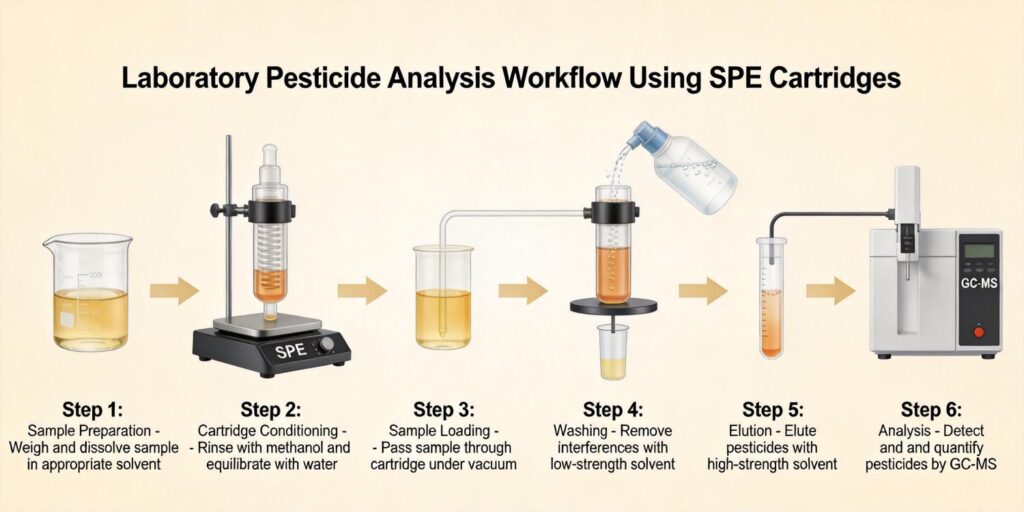
Requirements for Multi-Residue Pesticide Analysis
Multi-residue pesticide screening represents one of the most challenging analytical tasks in environmental and food safety testing. According to established literature, comprehensive screening workflows must address several critical requirements simultaneously. The primary objective is to detect and quantify a broad spectrum of pesticide residues at trace levels, typically in the parts-per-billion (ppb) to parts-per-trillion (ppt) range, across diverse sample matrices including water, soil, food products, and biological tissues.
Key requirements include:
- Broad analyte coverage: Methods must capture pesticides with varying chemical properties including organochlorines, organophosphates, carbamates, triazines, and newer chemical classes
- High sensitivity: Detection limits must meet regulatory requirements, often requiring enrichment factors of 100-1000x
- Matrix cleanup: Effective removal of interfering compounds such as humic acids, proteins, lipids, and pigments
- Compatibility with detection systems: Extracts must be suitable for LC-MS/MS, GC-MS, or other analytical platforms
- High throughput: Ability to process multiple samples efficiently, particularly for monitoring programs
Research by Font et al. (1993) demonstrated that solid-phase extraction in multi-residue pesticide analysis of water requires careful consideration of these factors to achieve reliable results across diverse analyte classes.
Challenges with Diverse Analyte Polarities
The chemical diversity of modern pesticides presents significant challenges for extraction method development. Pesticides span an enormous range of polarities, with log P values ranging from highly polar compounds like glyphosate (log P ~ -3.2) to highly non-polar compounds like DDT (log P ~ 6.9). This polarity range creates fundamental difficulties in developing a single extraction method that provides adequate recovery for all target analytes.
Specific challenges include:
- Hydrophilic compounds: Highly polar pesticides often exhibit poor retention on traditional reversed-phase sorbents, leading to breakthrough during sample loading
- Hydrophobic compounds: Non-polar pesticides may be difficult to elute from sorbents without strong organic solvents that could damage analytical columns
- Ionizable compounds: Many pesticides contain acidic or basic functional groups whose retention characteristics change dramatically with pH
- Matrix effects: Co-extracted matrix components can interfere with both extraction efficiency and subsequent analysis
Studies by Liska et al. (1990) comparing sorbents for solid-phase extraction of polar compounds from water highlighted the limitations of single-sorbent approaches for broad-spectrum pesticide analysis.
Sorbent Chemistry Selection for Broad Analyte Coverage
Selecting appropriate sorbent chemistry is critical for successful multi-residue pesticide screening. The choice depends on the specific analyte properties and sample matrix. Several sorbent options are available, each with distinct advantages:
Reversed-Phase Sorbents (C18, C8, HLB)
Traditional reversed-phase sorbents like C18 provide excellent retention for non-polar to moderately polar compounds. Hydrophilic-Lipophilic Balance (HLB) sorbents, containing both hydrophilic N-vinylpyrrolidone and lipophilic divinylbenzene monomers, offer enhanced retention for polar compounds while maintaining good recovery of non-polar analytes. HLB sorbents are particularly valuable for multi-residue methods as they don’t require conditioning and can retain compounds across a wide polarity range.
Mixed-Mode Sorbents (MCX, MAX, WCX, WAX)
Mixed-mode sorbents combine reversed-phase retention with ion-exchange functionality, providing orthogonal selectivity. Cation-exchange sorbents (MCX, WCX) are effective for basic compounds, while anion-exchange sorbents (MAX, WAX) target acidic analytes. These sorbents allow selective retention based on both hydrophobicity and ionic interactions, enabling cleaner extracts and better recovery of ionizable pesticides.
Specialty Sorbents
For specific applications, specialty sorbents may be appropriate:
- Florisil: Effective for cleanup of organochlorine pesticides from fatty matrices
- Graphitized carbon black: Useful for removing pigments and other planar compounds
- Polymeric sorbents: Offer higher capacity and better pH stability than silica-based materials
Research by Pichon et al. (1996) demonstrated that polymeric sorbents could simultaneously extract polar acidic, neutral, and basic pesticides while effectively removing humic and fulvic acid interferences.
Optimized Conditioning and Loading Steps
Proper conditioning and sample loading are essential for reproducible and efficient extraction. The conditioning step activates the sorbent and prepares it for optimal interaction with target analytes.
Conditioning Protocol
For most sorbents, conditioning involves sequential solvent steps:
- Solvation: Typically 3-5 mL of methanol or acetonitrile to wet the sorbent surface
- Equilibration: 3-5 mL of water or buffer matching the sample matrix to remove excess organic solvent
Critical considerations include:
- Flow rate: Should be controlled to ensure complete wetting (typically 1-5 mL/min)
- Drying avoidance: Sorbent should not be allowed to dry between conditioning and sample loading
- pH adjustment: For ionizable compounds, sample pH should be adjusted 2 units away from the pKa to ensure proper ionization state
Sample Loading Optimization
Sample loading parameters significantly impact recovery:
- Flow rate: Typically 1-10 mL/min, with slower rates improving retention of poorly retained compounds
- Sample volume: Must be optimized based on breakthrough volume studies
- Matrix modification: Addition of organic solvent (typically 5-10%) can improve recovery of hydrophobic compounds
- pH control: Critical for ionizable compounds to ensure proper retention mechanism
Studies by Mayer and Poole (1994) identified that procedural steps during conditioning and loading significantly affect recovery of semi-volatile compounds in SPE.
Sequential Washing Strategies
Effective washing removes interfering compounds while retaining target analytes. Sequential washing strategies typically employ solvents of increasing elution strength.
Water-Based Washes
Initial washes with water or aqueous buffers remove highly polar interferences:
- Deionized water: Removes salts and very polar compounds
- pH-adjusted water: Can selectively remove acidic or basic interferences
- Low-percentage organic: 5-10% methanol or acetonitrile in water removes moderately polar interferences
Organic Washes
Subsequent washes with organic solvents remove less polar interferences:
- Moderate organic: 20-40% methanol or acetonitrile in water
- Pure organic: 100% methanol, acetonitrile, or ethyl acetate for specific applications
- pH-adjusted organic: Organic solvents with acid or base modifiers for selective washing
Drying Step
After washing, a drying step (typically 5-30 minutes under vacuum) removes residual water that could interfere with elution or subsequent analysis.
Research by de Zeeuw and Franke (2000) in their comprehensive SPE procedure for broad-spectrum drug screening emphasized the importance of sequential washing strategies for achieving clean extracts.
Elution Solvents Compatible with LC-MS/MS
Elution solvent selection must balance complete analyte recovery with compatibility with downstream analytical techniques, particularly LC-MS/MS.
Solvent Properties
Ideal elution solvents for LC-MS/MS compatibility should:
- Provide complete elution of all target analytes
- Be miscible with aqueous mobile phases
- Have low volatility for concentration steps
- Exhibit minimal ion suppression in MS detection
- Be compatible with analytical column chemistry
Common Elution Solvents
Several solvent systems have proven effective:
- Methanol: Excellent elution strength, good MS compatibility, but can cause excessive matrix co-extraction
- Acetonitrile: Strong elution power, excellent MS compatibility, lower background in UV detection
- Acetone: Very strong eluent, but may cause issues in LC-MS due to high volatility
- Ethyl acetate: Good for non-polar compounds, but immiscible with water requiring evaporation
- Mixed solvents: Combinations like methanol:acetonitrile (50:50) or methanol:ethyl acetate (50:50) can provide balanced properties
pH-Modified Elution
For mixed-mode sorbents, pH-modified elution is essential:
- Acidic elution: For basic compounds from cation-exchange sorbents (e.g., 5% formic acid in methanol)
- Basic elution: For acidic compounds from anion-exchange sorbents (e.g., 5% ammonium hydroxide in methanol)
Studies by Bowers et al. (1997) demonstrated that automated SPE with tandem MS quantification at picogram levels requires careful optimization of elution solvents for compatibility with mass spectrometric detection.
Performance Metrics: Recovery, Matrix Effects, Reproducibility
Comprehensive method validation requires evaluation of multiple performance metrics to ensure reliable results.
Recovery Studies
Recovery should be evaluated across the entire method, including:
- Absolute recovery: Comparison of extracted samples to neat standards
- Relative recovery: Comparison to matrix-matched standards
- Acceptance criteria: Typically 70-120% recovery with RSD < 20%
- Concentration dependence: Recovery should be consistent across the calibration range
Matrix Effects Evaluation
Matrix effects, particularly ion suppression/enhancement in LC-MS/MS, must be quantified:
- Post-extraction addition: Compare response of standards added to extracted matrix vs. neat standards
- Matrix factor: Ratio of response in matrix vs. neat solution
- Mitigation strategies: Include matrix-matched calibration, isotope-labeled internal standards, improved cleanup
Reproducibility Assessment
Method reproducibility should be evaluated at multiple levels:
- Intra-day precision: Multiple replicates within a single batch
- Inter-day precision: Multiple batches over several days
- Intermediate precision: Different analysts, instruments, or laboratories
- Robustness: Response to small, deliberate variations in method parameters
Additional Validation Parameters
Complete method validation should also include:
- Linearity: Typically over 2-3 orders of magnitude
- Limit of detection/quantification: Based on signal-to-noise ratios
- Selectivity/specificity: Ability to distinguish target analytes from interferences
- Carryover: Assessment of contamination between samples
Research by Simpson et al. (1998) on high-throughput bioanalysis using 96-well SPE plates demonstrated that rigorous validation of recovery, matrix effects, and reproducibility is essential for reliable multi-residue screening methods.
Conclusion
Designing effective SPE workflows for multi-residue pesticide screening requires careful consideration of diverse analyte properties, appropriate sorbent selection, optimized procedural steps, and comprehensive method validation. The complexity of modern pesticide analysis demands sophisticated approaches that balance broad analyte coverage with clean extracts and compatibility with sensitive detection techniques like LC-MS/MS. By systematically addressing each component of the SPE workflow—from sorbent chemistry to final validation—analysts can develop robust methods that meet the stringent requirements of regulatory monitoring and food safety testing.
For laboratories seeking to implement or optimize multi-residue pesticide screening methods, Poseidon Scientific offers a comprehensive range of SPE products including HLB cartridges, MAX mixed-mode sorbents, MCX cation-exchange materials, and 96-well SPE plates for high-throughput applications. These products are designed to address the specific challenges of multi-residue analysis while providing the reproducibility and performance required for regulatory compliance.