Environmental Concern of Perchlorate Contamination
Perchlorate contamination in drinking water has emerged as a significant environmental and public health concern over the past two decades. Perchlorate (ClO4–) is a highly soluble and stable anion that originates primarily from industrial activities, particularly the manufacture and use of rocket propellants, fireworks, and explosives. Unlike many other contaminants, perchlorate does not readily degrade in the environment and can persist in groundwater for decades, creating long-term exposure risks.
The primary health concern associated with perchlorate exposure is its ability to competitively inhibit iodide uptake by the thyroid gland. This interference can disrupt thyroid hormone production, potentially leading to developmental issues in fetuses and infants, as well as metabolic disorders in adults. Regulatory agencies worldwide have established stringent limits for perchlorate in drinking water, with the U.S. Environmental Protection Agency (EPA) setting a maximum contaminant level (MCL) of 15 parts per billion (ppb) and some states implementing even lower limits.
Environmental monitoring for perchlorate presents unique analytical challenges due to its high solubility, low molecular weight, and the need for detection at trace levels in complex water matrices. Traditional sample preparation methods often lack the specificity and sensitivity required for reliable perchlorate determination, making solid-phase extraction (SPE) an essential tool for environmental laboratories.
Sample Collection and Storage Procedures
Proper sample collection and storage are critical for accurate perchlorate analysis in drinking water. Environmental samples should be collected in clean, high-density polyethylene (HDPE) or glass containers that have been thoroughly rinsed with deionized water. Plastic containers should be avoided if the analysis will involve organic solvent extraction, as plasticizers may interfere with the analysis.
For perchlorate analysis, samples should be collected without headspace to minimize volatilization and stored at 4°C immediately after collection. The addition of preservatives is generally not required for perchlorate analysis, but samples should be analyzed within 14 days of collection to ensure stability. If longer storage is necessary, samples can be preserved by acidification to pH < 2 with hydrochloric acid and stored at 4°C for up to 28 days.
Field blanks and quality control samples should be collected following the same procedures to monitor for potential contamination during sampling and transport. The sample volume required depends on the expected perchlorate concentration and the detection method, but typically 100-500 mL is sufficient for SPE concentration followed by ion chromatography or LC-MS analysis.
MAX Cartridge Chemistry and Anion-Exchange Mechanism
The MAX (Mixed-mode Anion eXchange) SPE cartridge represents a sophisticated approach to perchlorate extraction that combines multiple retention mechanisms for enhanced selectivity. Unlike traditional anion exchange sorbents, MAX cartridges incorporate both hydrophobic and anion exchange functionalities in a single polymeric matrix.
The chemistry of MAX sorbents typically involves a polymeric backbone (often divinylbenzene/N-vinylpyrrolidone copolymer) functionalized with quaternary ammonium groups. This dual-mode design provides several advantages for perchlorate extraction:
- Hydrophobic interactions from the polymeric backbone retain non-polar interferences
- Strong anion exchange through quaternary ammonium groups selectively retains perchlorate ions
- pH-independent retention of anions due to the permanent positive charge of quaternary ammonium groups
The anion exchange mechanism operates through electrostatic interactions between the positively charged quaternary ammonium sites on the sorbent and the negatively charged perchlorate ions in solution. Perchlorate, being a large, poorly hydrated anion with high charge density, exhibits particularly strong affinity for anion exchange sites, allowing for selective retention even in the presence of competing anions like chloride, sulfate, and nitrate.
This mixed-mode approach significantly improves selectivity compared to traditional reversed-phase or single-mode anion exchange sorbents, reducing matrix effects and improving method robustness for complex water samples.
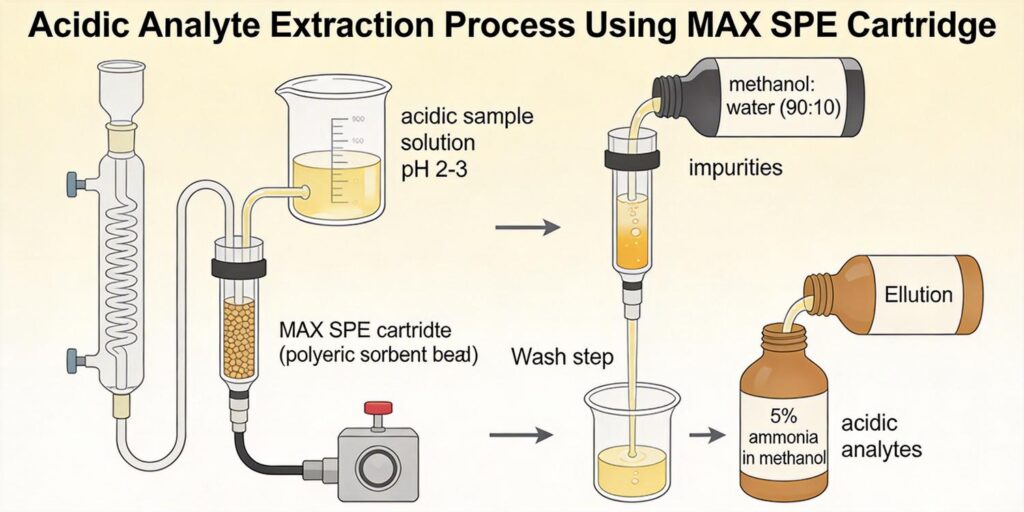
Conditioning with Methanol and Water
Proper conditioning of MAX cartridges is essential for optimal perchlorate recovery and reproducible results. The conditioning process serves three primary purposes: activation of the sorbent, removal of potential interferences, and establishment of the appropriate chemical environment for sample loading.
The standard conditioning protocol for MAX cartridges involves sequential solvent treatments:
- Methanol conditioning: 3-5 mL of HPLC-grade methanol is passed through the cartridge to solvate the polymeric backbone and ensure complete wetting of the hydrophobic domains. This step activates the sorbent by opening the polymer structure and making the anion exchange sites accessible.
- Water conditioning: 3-5 mL of deionized water (pH adjusted to match the sample pH) is passed through the cartridge to remove methanol and establish an aqueous environment compatible with the water sample. This step is critical to prevent sample precipitation or phase separation during loading.
During conditioning, flow rates should be maintained at 1-2 mL/min to ensure thorough solvent penetration without creating channeling in the sorbent bed. The cartridge should not be allowed to dry between conditioning and sample loading, as this can reduce retention efficiency and cause poor reproducibility.
For trace-level perchlorate analysis, additional conditioning steps may include washing with dilute acid or base solutions to remove any residual contaminants from the manufacturing process, though certified clean sorbents like those from Poseidon Scientific typically require minimal additional cleanup.
Sample Loading at Neutral pH to Retain Perchlorate Ions
Sample loading represents the most critical step in the MAX SPE method for perchlorate extraction. The loading conditions must be optimized to maximize perchlorate retention while minimizing the retention of interfering anions.
For MAX cartridges, sample loading should be performed at neutral to slightly basic pH (pH 7-8). At this pH range:
- Perchlorate exists predominantly as the fully dissociated anion (ClO4–)
- The quaternary ammonium groups on the MAX sorbent maintain their positive charge
- Competing anions like bicarbonate and carbonate are present in their anionic forms but have lower affinity for the anion exchange sites
The sample loading flow rate should be carefully controlled to ensure adequate contact time between the perchlorate ions and the anion exchange sites. Typical flow rates range from 1-5 mL/min, depending on the sorbent mass and cartridge configuration. For 60 mg MAX cartridges, a flow rate of 2-3 mL/min provides optimal retention for 100-500 mL sample volumes.
During loading, the sample should be passed through the cartridge completely without allowing the sorbent bed to dry. If the sample contains high levels of particulate matter, filtration through a 0.45 μm membrane filter is recommended prior to SPE to prevent cartridge clogging and ensure consistent flow rates.
The loading capacity of MAX cartridges for perchlorate is typically in the range of 1-2 mg per 60 mg of sorbent, which is more than sufficient for environmental water samples where perchlorate concentrations rarely exceed 100 μg/L.
Washing with Water to Remove Competing Ions
Following sample loading, a washing step is essential to remove weakly retained matrix components while maintaining perchlorate retention on the cartridge. The washing protocol must strike a balance between effective cleanup and minimal perchlorate loss.
For MAX cartridges, deionized water is typically used as the wash solvent. The water wash serves several important functions:
- Removal of inorganic salts: Chloride, nitrate, and sulfate ions, which have lower affinity for the anion exchange sites than perchlorate, are effectively removed by water washing
- Elimination of polar organic compounds: Hydrophilic organic acids and other polar compounds that may be retained through weak interactions are washed away
- Reduction of matrix effects: By removing competing ions, the water wash minimizes potential interferences in subsequent chromatographic analysis
The standard washing protocol involves passing 3-5 mL of deionized water through the cartridge at a flow rate of 1-2 mL/min. The wash volume should be optimized based on the specific water matrix, as samples with high ionic strength may require larger wash volumes.
It is important to note that perchlorate, with its high affinity for quaternary ammonium sites, remains strongly retained during the water wash. However, excessive washing or the use of organic solvents in the wash step should be avoided, as these can potentially elute perchlorate or compromise the anion exchange mechanism.
After washing, the cartridge may be dried briefly under vacuum (5-10 minutes) to remove residual water before elution, though complete drying is not necessary and may actually reduce elution efficiency for some applications.
Elution Using Ammonium Hydroxide in Methanol
The elution step represents the final critical phase in the MAX SPE method, where retained perchlorate is selectively recovered from the cartridge. The choice of elution solvent must satisfy two key requirements: effectively disrupt the electrostatic interactions between perchlorate and the anion exchange sites, and provide compatibility with subsequent analytical techniques.
For MAX cartridges, the most effective elution solvent for perchlorate is ammonium hydroxide in methanol, typically at concentrations of 2-5% (v/v). This eluent combination works through multiple mechanisms:
- Ion exchange competition: The hydroxide ions (OH–) compete with perchlorate for the anion exchange sites
- Ion pairing disruption: Ammonium ions can form ion pairs with perchlorate, reducing its affinity for the stationary phase
- Solvent strength: Methanol provides sufficient solvent strength to elute perchlorate while maintaining compatibility with most analytical methods
The standard elution protocol involves passing 2-3 mL of 5% ammonium hydroxide in methanol through the cartridge at a slow flow rate (0.5-1 mL/min) to ensure complete perchlorate recovery. The eluent volume should be optimized based on the specific cartridge size and sorbent mass, with 60 mg MAX cartridges typically requiring 2 mL for quantitative recovery.
For maximum recovery, the elution step may be performed in two aliquots: an initial 1 mL elution followed by a second 1 mL elution. The combined eluates are then typically evaporated to dryness under a gentle stream of nitrogen and reconstituted in a smaller volume of water or mobile phase compatible with the detection method.
Alternative elution solvents include potassium hydroxide in methanol or tetrabutylammonium hydroxide, but ammonium hydroxide is generally preferred due to its volatility (facilitating solvent evaporation) and compatibility with both ion chromatography and LC-MS systems.
Ion Chromatography or LC-MS Detection
Following SPE concentration and cleanup, perchlorate is typically quantified using either ion chromatography (IC) or liquid chromatography-mass spectrometry (LC-MS). Each technique offers distinct advantages for perchlorate analysis in drinking water.
Ion Chromatography (IC) Detection
Ion chromatography with conductivity detection remains the most widely used method for perchlorate analysis in environmental laboratories. The EPA Method 314.0 specifically addresses perchlorate determination by IC and recommends the following conditions:
- Column: High-capacity anion exchange column (e.g., Dionex IonPac AS16 or equivalent)
- Mobile phase: Sodium hydroxide gradient (typically 10-100 mM)
- Detection: Suppressed conductivity detection
- Detection limit: 0.5-1.0 μg/L with direct injection; sub-μg/L with preconcentration
The main advantage of IC is its specificity for ionic compounds and excellent sensitivity for perchlorate. However, IC can suffer from interferences from other anions that co-elute with perchlorate, particularly in complex water matrices.
Liquid Chromatography-Mass Spectrometry (LC-MS) Detection
LC-MS methods, particularly using tandem mass spectrometry (LC-MS/MS), offer superior specificity and lower detection limits for perchlorate analysis. EPA Method 331.0 and 332.0 describe LC-MS methods for perchlorate with the following typical conditions:
- Column: Reversed-phase C18 or polar-embedded stationary phase
- Mobile phase: Ammonium acetate or formate in water/methanol
- Ionization: Electrospray ionization in negative mode
- Detection: Selected reaction monitoring (SRM) of m/z 99→83 transition
- Detection limit: 0.02-0.05 μg/L
LC-MS/MS provides unambiguous identification of perchlorate through its characteristic mass transitions and isotope patterns, making it the method of choice for regulatory compliance monitoring and research applications requiring the highest confidence in results.
Method Selection Considerations
The choice between IC and LC-MS depends on several factors:
- Required detection limits: LC-MS offers lower detection limits (ng/L range)
- Matrix complexity: LC-MS provides better specificity in complex matrices
- Instrument availability: IC systems are more widely available in environmental labs
- Throughput requirements: IC typically offers faster analysis times
- Regulatory requirements: Specific methods may be mandated for compliance monitoring
Regardless of the detection method, the MAX SPE procedure described above provides excellent sample cleanup and concentration, enabling reliable perchlorate determination at levels well below regulatory limits. The combination of MAX SPE with either IC or LC-MS represents a robust, validated approach for perchlorate monitoring in drinking water that meets the stringent requirements of environmental regulatory agencies worldwide.
For laboratories implementing this method, regular quality control measures including method blanks, matrix spikes, duplicate analyses, and continuing calibration verification are essential to ensure data quality and regulatory compliance. The use of certified reference materials and participation in proficiency testing programs further validate method performance and laboratory competency.