Chemical Profile of Organic Acids in Bioanalysis
Organic acids represent a diverse class of compounds that play crucial roles in biological systems, including metabolic intermediates, signaling molecules, and biomarkers of disease states. In bioanalysis, these compounds typically feature carboxylic acid functional groups with pKa values ranging from 2-8, classifying them as weak acids. Common examples include citric acid, lactic acid, succinic acid, fumaric acid, and various fatty acids.
From a chemical perspective, organic acids exist in equilibrium between their protonated (neutral) and deprotonated (anionic) forms depending on the pH of their environment. At physiological pH (approximately 7.4), most organic acids are predominantly in their anionic form, making them highly polar and water-soluble. This characteristic presents significant challenges for traditional reversed-phase extraction methods, which typically require analytes to be in their neutral form for effective retention.
The analytical detection of organic acids in biological fluids is further complicated by their typically low concentrations (often in the ng/mL to μg/mL range), complex matrix interferences from endogenous compounds, and the presence of structurally similar isomers. Common biological matrices for organic acid analysis include plasma, serum, urine, cerebrospinal fluid, and tissue homogenates, each presenting unique challenges in sample preparation.
Why Strong Anion Exchange (MAX) Provides High Retention
Mixed-mode Anion eXchange (MAX) sorbents represent a breakthrough in solid-phase extraction technology specifically designed for acidic compounds. Unlike traditional reversed-phase materials, MAX sorbents combine two distinct retention mechanisms: reversed-phase hydrophobic interactions and strong anion exchange. This dual-mode approach provides superior retention for organic acids compared to single-mode sorbents.
The strong anion exchange component of MAX sorbents typically features quaternary ammonium groups that maintain a permanent positive charge across the entire pH range. This characteristic is particularly advantageous for organic acid extraction because:
- pH-Independent Retention: Unlike weak anion exchangers that require specific pH conditions to maintain their charge, MAX sorbents remain positively charged regardless of pH, ensuring consistent retention of anionic analytes.
- Enhanced Selectivity: The anion exchange mechanism provides orthogonal selectivity to reversed-phase interactions, allowing for cleaner extracts by selectively retaining acidic compounds while excluding neutral and basic interferences.
- High Capacity: Modern MAX sorbents feature tightly controlled ion-exchange capacities (typically around 0.25 meq/g), ensuring reproducible extraction protocols and high loading capacities for trace-level analytes.
- Reduced Matrix Effects: The mixed-mode retention minimizes non-specific binding and silanol interactions that can complicate method development with traditional silica-based sorbents.
Research indicates that MAX sorbents provide significantly higher recoveries for polar acidic compounds compared to conventional C18 or HLB phases, particularly for analytes with pKa values between 2-8. The water-wettable polymeric backbone of modern MAX sorbents ensures complete wetting and consistent flow characteristics, even with highly aqueous samples.
Sample Pretreatment and pH Adjustment
Proper sample pretreatment is critical for successful MAX SPE extraction of organic acids from biological fluids. The primary objectives of pretreatment include protein removal, matrix simplification, and pH adjustment to optimize analyte retention.
Protein Precipitation
For plasma and serum samples, protein precipitation represents the most common initial step. Organic solvents such as acetonitrile or methanol are typically added at a 2:1 or 3:1 (solvent:sample) ratio. This approach offers several advantages:
- Simultaneous protein precipitation and analyte extraction
- Reduction of phospholipid content (a major source of matrix effects in LC-MS)
- Compatibility with subsequent SPE procedures
After precipitation, samples should be centrifuged at 10,000-15,000 × g for 10-15 minutes, and the supernatant carefully transferred to avoid disturbing the protein pellet.
pH Adjustment Strategy
pH adjustment serves two critical functions in MAX SPE workflows:
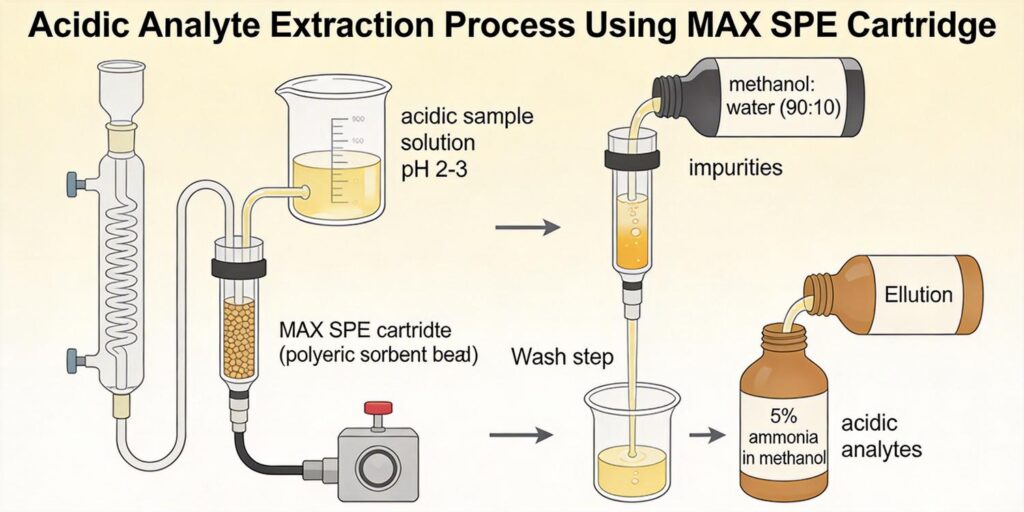
- Analyte Ionization Control: Organic acids must be in their anionic form for effective retention on MAX sorbents. For compounds with pKa values between 2-8, adjusting the sample pH to at least 2 units above the highest pKa ensures complete deprotonation. Typically, samples are adjusted to pH 8-9 using ammonium hydroxide or appropriate buffer solutions.
- Interference Minimization: Basic and neutral compounds remain uncharged or positively charged at alkaline pH, minimizing their retention on MAX sorbents and reducing matrix interferences.
For urine samples, dilution with an appropriate buffer (typically 1:1 to 1:4 dilution) helps reduce ionic strength and minimize competitive binding from endogenous anions. The optimal dilution factor should be determined empirically based on analyte concentration and matrix complexity.
Conditioning and Loading Procedures
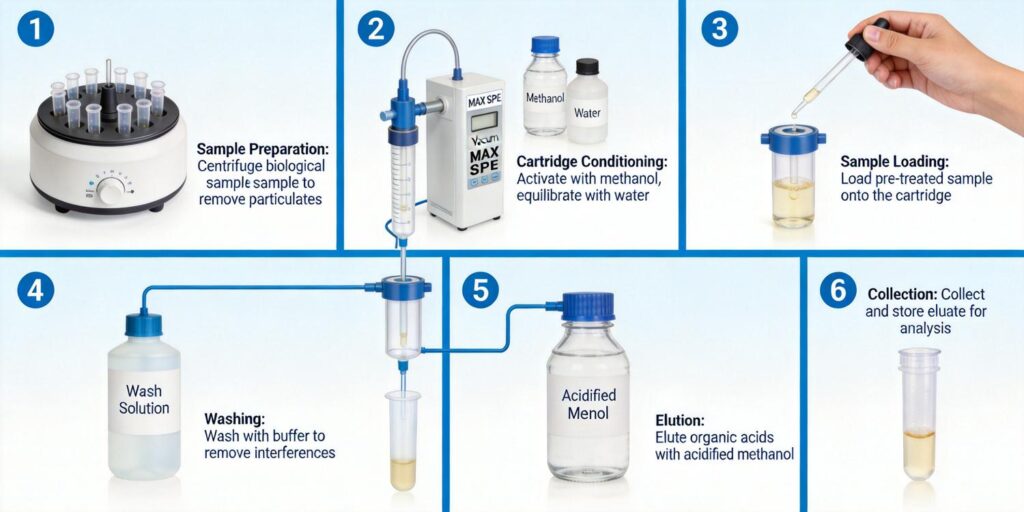
Cartridge Conditioning
Proper conditioning of MAX cartridges is essential for achieving reproducible recoveries and consistent flow characteristics. The standard conditioning protocol includes:
- Solvent Activation: 2-3 mL of methanol or acetonitrile to solvate the polymeric backbone and ensure complete wetting of the sorbent bed.
- Equilibration: 2-3 mL of water or aqueous buffer (pH 8-9) to create an aqueous environment compatible with sample loading.
It is crucial to prevent the sorbent bed from drying out between conditioning and sample loading, as this can lead to poor recovery and inconsistent results. The flow rate during conditioning should be maintained at 1-2 mL/min to ensure complete solvent exchange.
Sample Loading Optimization
Sample loading represents the most critical step in MAX SPE workflows. Several factors must be optimized:
- Flow Rate Control: Loading flow rates should be maintained at 1-2 mL/min to ensure adequate interaction time between analytes and sorbent. Excessive flow rates can lead to breakthrough and reduced recovery.
- Sample Volume: The optimal loading volume depends on analyte concentration, sorbent capacity, and matrix complexity. For trace-level analysis, larger volumes (up to 10-20 mL) may be loaded to achieve adequate preconcentration.
- pH Monitoring: The pH of the effluent should be monitored during loading to ensure it remains within the optimal range (pH 8-9). Significant pH drops may indicate insufficient buffer capacity or excessive acid content in the sample.
For complex biological matrices, loading in multiple aliquots with intermittent mixing can improve recovery by preventing localized overloading and ensuring uniform distribution of analytes throughout the sorbent bed.
Washing with Organic Solvents to Remove Lipids
Effective washing protocols are essential for removing non-polar interferences while retaining target organic acids. The dual retention mechanism of MAX sorbents allows for aggressive washing conditions that would typically elute analytes from single-mode phases.
Primary Wash: Aqueous Buffer
The initial wash typically employs 2-3 mL of the same buffer used for sample pH adjustment (pH 8-9). This step removes:
- Residual proteins and peptides
- Highly polar neutral compounds
- Excess salts and buffer components
The aqueous wash should contain 5-10% organic solvent (methanol or acetonitrile) to maintain sorbent wettability and prevent channel formation.
Secondary Wash: Organic Solvent
The critical washing step for lipid removal involves 2-3 mL of methanol or acetonitrile. At this stage:
- Lipid Removal: Neutral lipids, phospholipids, and other non-polar interferences are effectively eluted due to their reversed-phase retention characteristics.
- Analyte Retention: Organic acids remain strongly retained through ionic interactions with the quaternary ammonium groups, unaffected by the organic solvent.
- Drying Step: After organic washing, cartridges should be dried under vacuum (5-10 minutes at maximum vacuum) or positive pressure to remove residual water and organic solvent. Complete drying is essential for efficient elution in the subsequent step.
For particularly lipid-rich samples (e.g., plasma, tissue homogenates), additional washing with hexane or methyl tert-butyl ether (MTBE) may be incorporated to remove non-polar interferences more effectively.
Elution with Acidified Solvent Mixtures
Elution Mechanism
Elution of organic acids from MAX sorbents requires disruption of both ionic and hydrophobic interactions. This is typically achieved through a two-step approach:
- Ionic Bond Disruption: Acidification of the elution solvent protonates the carboxylic acid groups, neutralizing their negative charge and breaking the ionic interaction with the quaternary ammonium sites.
- Hydrophobic Interaction Disruption: Organic solvents with appropriate elution strength overcome the reversed-phase retention of the neutralized acids.
Optimized Elution Protocol
The standard elution protocol for MAX sorbents involves:
- Acidified Organic Solvent: 2-3 mL of methanol or acetonitrile containing 2-5% formic acid. The acid concentration should be optimized based on analyte pKa values and recovery requirements.
- Collection Strategy: Eluates should be collected in silanized glass vials or polypropylene tubes to minimize analyte adsorption. For maximum recovery, two sequential elutions with 1-1.5 mL each are often more effective than a single large-volume elution.
- Evaporation and Reconstitution: Eluates are typically evaporated to dryness under a gentle stream of nitrogen at 30-40°C. Reconstitution in mobile phase-compatible solvent (typically 50-100 μL) provides both concentration and solvent exchange benefits.
For particularly polar organic acids or those with multiple carboxylic acid groups, alternative elution strategies may include:
- Acidified methanol/water mixtures (e.g., 90:10 methanol:water with 2% formic acid)
- Ammonium formate or acetate buffers in organic solvent
- Stepwise elution with increasing organic strength
LC-MS Validation Parameters for Organic Acid Assays
Method Validation Requirements
Comprehensive validation of MAX SPE-LC-MS methods for organic acid analysis should address the following parameters:
- Selectivity and Specificity: Demonstration of no interference from endogenous compounds at the retention times of target analytes and internal standards.
- Linearity: Typically evaluated over 2-3 orders of magnitude with correlation coefficients (r2) ≥ 0.99.
- Accuracy and Precision: Intra-day and inter-day precision should demonstrate ≤15% RSD at all concentration levels, with accuracy within ±15% of nominal values.
- Recovery: Extraction recovery should be determined at low, medium, and high concentration levels, typically targeting 70-120% with ≤15% RSD.
- Matrix Effects: Ion suppression/enhancement should be evaluated using post-extraction spiking approaches, with matrix factors ideally between 0.85-1.15.
- Stability: Assessment of analyte stability in biological matrices under various storage conditions and during sample processing.
LC-MS Method Optimization
For organic acid analysis, several LC-MS parameters require special attention:
- Chromatographic Separation: HILIC or reversed-phase chromatography with acidic mobile phases (typically 0.1% formic acid) provides optimal separation and ionization for organic acids.
- Ionization Mode: Negative electrospray ionization (ESI-) is generally preferred for organic acids due to their acidic nature and tendency to form stable deprotonated molecules [M-H]–.
- Mass Spectrometer Parameters: Optimized declustering potentials, collision energies, and source temperatures are critical for achieving adequate sensitivity and selectivity.
- Internal Standard Selection: Stable isotope-labeled analogs of target analytes represent the ideal internal standards, providing compensation for both extraction efficiency and matrix effects.
Quality Control Implementation
Robust quality control measures should include:
- Blank samples (extracted matrix without analytes)
- Zero samples (extracted matrix with internal standard only)
- Calibration standards covering the entire analytical range
- Quality control samples at low, medium, and high concentrations
- Carryover assessment samples
- System suitability tests
The MAX SPE workflow, when properly optimized and validated, provides a robust platform for the sensitive and selective analysis of organic acids in complex biological matrices. The combination of strong anion exchange and reversed-phase retention mechanisms offers superior cleanup and preconcentration capabilities compared to traditional extraction approaches, making it an essential tool in modern bioanalytical laboratories.