Structural Characteristics of Basic Pharmaceuticals
Basic pharmaceuticals represent a significant class of therapeutic compounds characterized by their ability to accept protons and form positively charged species. These compounds typically contain nitrogen-containing functional groups such as primary, secondary, or tertiary amines, as well as more complex structures like quaternary ammonium compounds. The defining feature of basic drugs is their pKa value greater than 7, meaning they exist predominantly in their protonated, cationic form at physiological pH (7.4).
Common structural motifs include:
- Tertiary amines: Found in many antidepressants and antipsychotics
- Secondary amines: Present in various antihistamines and analgesics
- Quaternary ammonium compounds: Characterized by permanent positive charge regardless of pH
- Heterocyclic nitrogen-containing structures: Including pyridines, imidazoles, and piperazines
The protonation state of these compounds is pH-dependent, which becomes crucial for their extraction from complex biological matrices like plasma. At lower pH values (typically below the pKa), these compounds exist in their protonated, cationic forms, making them ideal candidates for ion-exchange purification strategies.
Retention Mechanism of Weak Cation Exchange (WCX) Sorbents
Weak cation exchange sorbents operate through a sophisticated mixed-mode retention mechanism that combines both ion-exchange and reversed-phase interactions. According to Waters documentation, Oasis WCX sorbents were specifically developed to provide better sample preparation for strong bases and quaternary amines, with a tightly controlled ion-exchange capacity of 0.7 meq/g.
The retention mechanism involves:
- Ion-exchange interactions: Carboxylic acid groups on the sorbent surface (pKa ~4-5) interact with protonated basic compounds through electrostatic attraction
- Reversed-phase interactions: Hydrophobic moieties on the polymeric backbone provide secondary retention through van der Waals forces
- Mixed-mode synergy: The combination of both mechanisms enhances retention for all types of basic analytes, especially strong bases
This dual retention mechanism offers several advantages over traditional reversed-phase SPE:
- Enhanced selectivity for basic compounds over neutral and acidic interferences
- Improved retention of highly polar basic compounds that might otherwise wash through reversed-phase sorbents
- Greater capacity for basic analytes due to multiple interaction sites
- Reduced matrix effects in subsequent LC-MS analysis
Adjusting Plasma pH to Optimize Ionic Interactions
pH manipulation is the cornerstone of successful WCX SPE for basic pharmaceuticals. Plasma samples typically have a pH of approximately 7.4, at which most basic drugs exist in their protonated forms. However, optimal retention on WCX sorbents often requires further pH adjustment to ensure complete protonation and maximize ionic interactions.
The general strategy involves:
- Acidification: Adding 2-4% phosphoric acid or formic acid to plasma samples to achieve pH 2-3
- Dilution factor: Typically 1:1 to 1:4 dilution with acidified aqueous solution
- pH verification: Ensuring the final sample pH is at least 2 units below the pKa of the target basic compounds
Research by de Zeeuw and Franke demonstrated that adjusting the starting pH to 2.2 significantly improved recoveries of polar acidic and neutral drugs in comprehensive SPE procedures. For basic compounds, this acidic environment ensures complete protonation and optimal retention on WCX sorbents.
Critical considerations include:
- Maintaining consistent pH across all samples for reproducible results
- Avoiding excessive dilution that might reduce retention of polar compounds
- Using volatile acids (formic, acetic) when compatibility with LC-MS is required
- Considering protein precipitation effects at extreme pH values
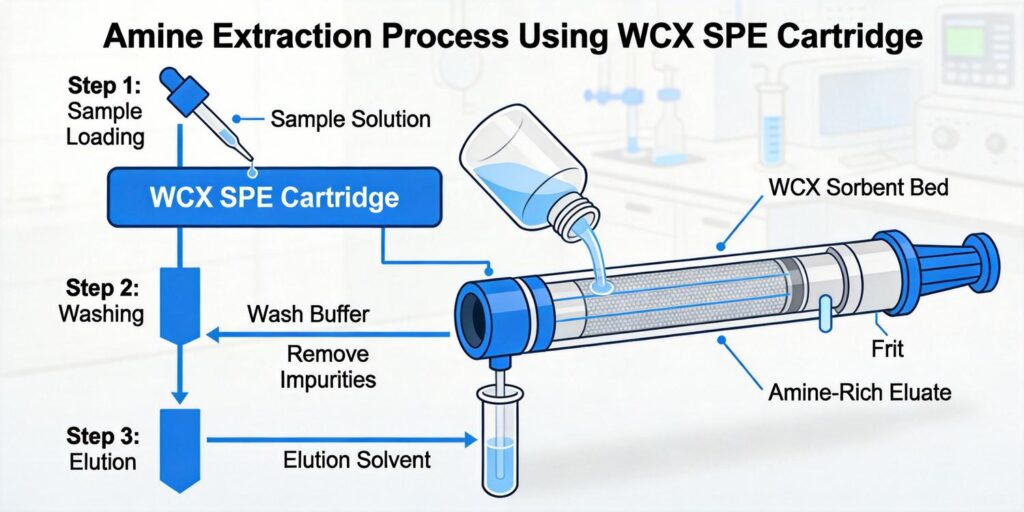
Cartridge Conditioning and Equilibration
Proper conditioning of WCX cartridges is essential for achieving optimal and reproducible results. The conditioning process serves multiple purposes: activating the sorbent, removing potential contaminants, and creating the appropriate chemical environment for analyte retention.
The standard conditioning protocol for WCX cartridges includes:
- Methanol activation: 2-3 mL of methanol to wet the hydrophobic polymer and remove any storage solvents
- Water rinse: 2-3 mL of deionized water to remove methanol and prepare for aqueous sample loading
- Conditioning with acidified solution: 2-3 mL of the same acidic solution used for sample preparation (e.g., 2% formic acid) to protonate the carboxylic acid groups on the sorbent
Key technical considerations:
- Never allow the sorbent bed to dry out between conditioning and sample loading
- Maintain a consistent flow rate (typically 1-2 mL/min) throughout the conditioning process
- Use sufficient solvent volumes to ensure complete conditioning (typically 2-3 bed volumes)
- For automated systems, ensure proper vacuum or pressure settings to achieve consistent flow
According to forensic SPE protocols, proper conditioning ensures that both the ion-exchange sites and reversed-phase domains are optimally prepared for analyte retention.
Wash Solutions to Remove Lipids and Proteins
Effective washing steps are crucial for removing matrix interferences while retaining target basic compounds. WCX sorbents allow for selective washing strategies that exploit the mixed-mode retention mechanism.
Recommended wash sequences typically include:
- Acidic aqueous wash: 2-5% formic acid in water to remove polar neutral and acidic interferences while maintaining basic compounds in their protonated, retained state
- Organic wash: 20-100% methanol or acetonitrile to remove lipids and non-polar interferences through disruption of reversed-phase interactions
- Optional additional washes: Depending on the specific application, additional washes with hexane or other non-polar solvents may be employed
The Waters Oasis 2 × 4 Method Development Protocol recommends specific wash sequences for different compound classes. For basic compounds on WCX sorbents, the protocol suggests:
- Wash 1: 2% Formic acid
- Wash 2: 100% MeOH
Critical wash optimization parameters:
- pH control: Maintain acidic conditions during aqueous washes to keep basic compounds protonated
- Organic content: Balance between effective interference removal and analyte retention
- Volume optimization: Use sufficient volume to remove interferences without excessive analyte loss
- Flow rate: Moderate flow rates (1-2 mL/min) for optimal washing efficiency
Elution Strategies Using Basic Solvents
Elution of basic compounds from WCX sorbents requires disruption of both ionic and hydrophobic interactions. The most effective elution strategies involve using basic organic solvents that neutralize the charge on both the analyte and the sorbent.
Standard elution protocols include:
- Basic organic elution: 2-5% ammonium hydroxide in methanol or acetonitrile
- Alternative basic modifiers: Triethylamine, diethylamine, or other volatile amines in organic solvents
- Solvent composition: Typically 70-90% organic solvent with basic modifier
The Waters protocol specifies elution with “5% NH4OH in MeOH” for basic compounds on WCX sorbents. This basic environment serves multiple purposes:
- Neutralizes the carboxylic acid groups on the sorbent (raising them above their pKa)
- Deprotonates the basic analytes, eliminating ionic interactions
- Provides sufficient organic content to disrupt hydrophobic interactions
- Maintains volatility for subsequent evaporation and reconstitution
Optimization considerations:
- Base concentration: Typically 2-5% for complete elution without excessive solvent effects
- Elution volume: 2-3 bed volumes (typically 2-4 mL for 1 cc cartridges)
- Flow rate: Slow elution (0.5-1 mL/min) for maximum recovery
- Collection strategy: Fraction collection to monitor elution profile and maximize recovery
Case Example: Antidepressant Drug Quantitation
A practical application of WCX SPE for basic pharmaceutical purification involves the quantitation of tricyclic antidepressants (TCAs) in plasma samples. TCAs like amitriptyline, nortriptyline, imipramine, and desipramine are classic examples of basic drugs with pKa values ranging from 9-10.
Sample Preparation Protocol:
- Sample pretreatment: 1 mL plasma + 1 mL 4% phosphoric acid, vortex mix
- Cartridge conditioning: 2 mL methanol, 2 mL water, 2 mL 2% formic acid
- Sample loading: Apply acidified plasma at 1 mL/min
- Wash steps: 2 mL 2% formic acid, followed by 2 mL methanol
- Elution: 2 × 2 mL 5% ammonium hydroxide in methanol
- Evaporation and reconstitution: Evaporate to dryness under nitrogen, reconstitute in mobile phase
Method Performance:
- Recovery: Typically 85-95% for most TCAs
- Matrix effects: Significant reduction compared to protein precipitation methods
- Selectivity: Excellent removal of phospholipids and other matrix interferences
- Linearity: Typically 1-500 ng/mL with R2 > 0.995
LC-MS/MS Conditions:
- Column: CORTECS C18+ 2.1 × 50 mm, 1.6 μm
- Mobile phase: 0.1% formic acid in water/acetonitrile gradient
- Detection: Positive electrospray ionization with MRM transitions
This case example demonstrates how WCX SPE provides the necessary selectivity and sensitivity for accurate quantitation of basic pharmaceuticals in complex biological matrices. The mixed-mode retention mechanism specifically targets basic compounds while effectively removing matrix interferences that could compromise analytical performance.
For laboratories considering WCX SPE implementation, Poseidon Scientific offers a comprehensive range of WCX SPE cartridges in various formats and sorbent masses to accommodate different sample volumes and analyte concentrations. Our products are designed to provide consistent performance and reliable results for your basic pharmaceutical analysis needs.