1. Mycotoxin Contamination Overview
Mycotoxins represent a significant food safety concern in grain commodities worldwide. These toxic secondary metabolites produced by fungi such as Aspergillus, Fusarium, and Penicillium species can contaminate various grains including corn, wheat, rice, and barley. Among the most concerning mycotoxins are aflatoxins (B1, B2, G1, G2), ochratoxin A, fumonisins, deoxynivalenol, and zearalenone, each posing unique health risks ranging from carcinogenic effects to endocrine disruption.
According to regulatory standards, maximum permissible levels for mycotoxins in grains are strictly controlled. For instance, the European Commission establishes that for concentrations less than 1 μg/kg, recoveries should range between 50-120%, while for concentrations between 1-10 μg/kg, recoveries should vary between 70-110%. These stringent requirements necessitate highly sensitive and selective analytical methods capable of detecting trace levels while effectively removing complex matrix interferences.
The challenge in mycotoxin analysis lies in the complex grain matrix containing pigments, lipids, proteins, and carbohydrates that can interfere with detection and quantification. Effective sample preparation becomes critical for achieving reliable results in LC-MS/MS analysis, where matrix effects can significantly impact method sensitivity and accuracy.
2. Grinding and Extraction of Grain Samples
Proper sample homogenization is the foundation of reproducible mycotoxin analysis. Grain samples should be ground to a consistent particle size using appropriate milling equipment. Research indicates that particle size significantly impacts extraction efficiency, with finer particles generally providing better analyte recovery due to increased surface area.
For cornmeal analysis, studies have shown that different particle sizes (fine, medium, and coarse) can affect method performance. The grinding process should achieve uniform particle distribution to ensure representative sampling and consistent extraction. After grinding, samples are typically weighed into extraction vessels, with typical sample sizes ranging from 5-25 grams depending on the expected contamination levels and method sensitivity requirements.
The extraction solvent selection depends on the target mycotoxins’ polarity. Common extraction solvents include acetonitrile-water mixtures, methanol-water combinations, or acidified aqueous solutions. For multi-mycotoxin methods, acetonitrile-water (84:16, v/v) or methanol-water (80:20, v/v) with 1% acetic acid are frequently employed to ensure efficient extraction of both polar and non-polar compounds.
3. Solvent Extraction Step
The extraction process typically involves mixing the ground sample with the selected solvent system, followed by vigorous shaking, vortexing, or ultrasonication. The extraction time and temperature are optimized based on the target analytes and matrix characteristics. For many mycotoxins, extraction at room temperature with 30-60 minutes of shaking provides satisfactory recovery.
After extraction, the mixture is centrifuged or filtered to separate the liquid extract from solid particles. The resulting extract contains not only the target mycotoxins but also numerous matrix components including lipids, pigments, proteins, and carbohydrates. This crude extract requires further cleanup before instrumental analysis to prevent column fouling, ion suppression in MS detection, and interference with accurate quantification.
Research on aflatoxin extraction from cornmeal has demonstrated that vortex-assisted extraction techniques can reduce processing time and minimize solvent exposure compared to traditional column-based methods. This approach also eliminates variations resulting from packing processes, contributing to improved method reproducibility.
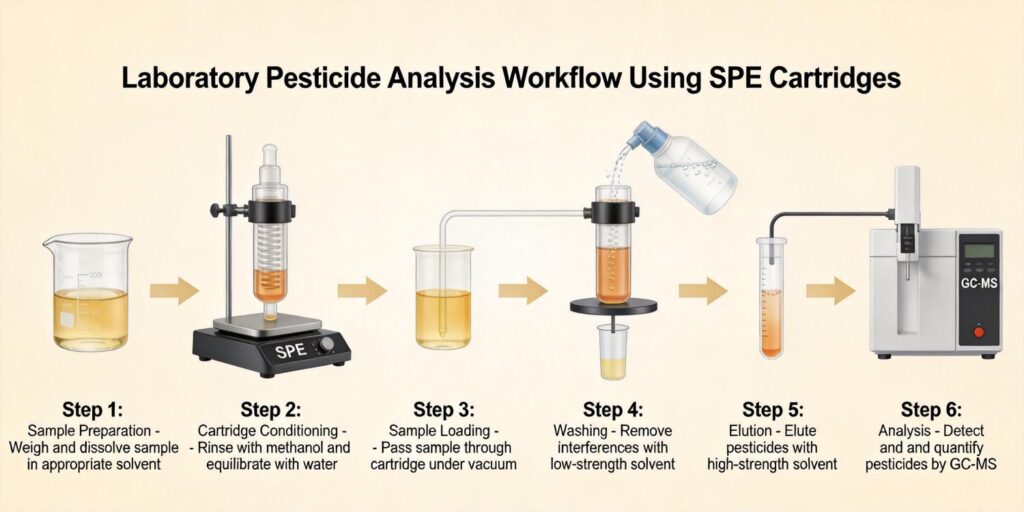
4. SPE Cleanup Workflow Using HLB Cartridges
Why HLB Cartridges for Mycotoxin Cleanup?
Hydrophilic-Lipophilic Balanced (HLB) cartridges, such as those from Poseidon Scientific’s product line, offer ideal characteristics for mycotoxin cleanup. The poly(divinylbenzene-co-N-vinylpyrrolidone) copolymer exhibits both hydrophilic and lipophilic retention properties, making it suitable for extracting medium-polar and non-polar organic compounds from aqueous-organic mixtures.
HLB sorbents are particularly effective for mycotoxin applications because they can retain analytes across a wide polarity range while effectively removing matrix interferences. The water-wettable nature of these polymeric sorbents eliminates the need for conditioning steps required with silica-based materials, simplifying the workflow and reducing solvent consumption.
Standard HLB Cleanup Protocol
The typical SPE cleanup procedure for mycotoxins in grain extracts involves the following optimized steps:
- Conditioning: 1 mL methanol followed by 1 mL water to prepare the sorbent bed
- Loading: 1 mL of appropriately diluted extract (typically 65% ethanol-water or similar organic-aqueous mixture)
- Washing: 1 mL of 5% methanol-water to remove salts and other polar interferences
- Elution: 1 mL of appropriate organic solvent (ethanol, acetonitrile, or methanol-based mixtures)
Studies have demonstrated that mycotoxins can be effectively retained on HLB cartridges using loading solutions containing 45-65% organic solvent. The optimal loading concentration depends on the specific mycotoxin polarity profile and should be determined during method development.
5. Removal of Pigments and Lipids
Grain matrices contain significant amounts of pigments (carotenoids, chlorophyll derivatives) and lipids that can interfere with chromatographic separation and mass spectrometric detection. HLB cartridges effectively remove these interferences through selective retention mechanisms.
The washing step plays a crucial role in pigment removal. A 5% methanol-water wash effectively removes polar pigments and salts while retaining mycotoxins on the sorbent. For more complex matrices, additional wash steps with slightly stronger solvents may be necessary, but care must be taken to avoid premature elution of target analytes.
Lipid removal is particularly important for LC-MS/MS analysis, as lipids can cause significant matrix effects and ion suppression. HLB sorbents have demonstrated excellent phospholipid removal capabilities, with studies showing removal of up to 95% of common matrix interferences including phospholipids. This cleanup efficiency is superior to protein precipitation methods and contributes to improved method sensitivity and reproducibility.
6. Elution Solvent Optimization
Solvent Selection Criteria
The choice of elution solvent significantly impacts mycotoxin recovery and method sensitivity. Several factors must be considered:
- Solvent strength: Must be sufficient to disrupt analyte-sorbent interactions
- Compatibility: Should be compatible with subsequent LC-MS/MS analysis
- Volatility: Important for solvent evaporation and concentration steps
- Toxicity: Operator safety considerations
Optimized Elution Conditions
Research comparing various elution solvents (tetrahydrofuran, acetonitrile, ethyl acetate, acetone, cyclohexane, ethanol, and methanol) has demonstrated that ethanol provides excellent recovery for many mycotoxins while offering favorable volatility and lower toxicity compared to other options. A typical optimized protocol uses 1 mL of ethanol for elution, providing high recovery with minimal solvent volume.
For multi-mycotoxin methods, solvent mixtures may be necessary. A 90:10 acetonitrile-methanol mixture has shown excellent elution efficiency for diverse mycotoxin classes. The elution volume should be optimized during method development, with studies indicating that 1 mL aliquots often provide satisfactory recovery while minimizing dilution effects.
Technical considerations include allowing the cartridge to soak with eluent for 0.5-1 minute before collection to improve recovery, and using several smaller eluent aliquots rather than one large volume when dealing with strongly retained analytes.
7. LC-MS/MS Analysis
Method Sensitivity and Validation
Following SPE cleanup, the eluate is typically evaporated to dryness and reconstituted in mobile phase compatible solvent for LC-MS/MS analysis. The cleanup efficiency directly impacts method sensitivity, with properly cleaned extracts showing significantly reduced matrix effects and improved detection limits.
Validation parameters for mycotoxin methods should include:
- Linearity: Typically demonstrated over 1-2 orders of magnitude
- Limit of Detection (LOD): Ranging from 0.01-0.32 ng/g for various mycotoxins
- Limit of Quantification (LOQ): Generally 3-10 times the LOD
- Recovery: 85-115% for most mycotoxins at relevant concentration levels
- Precision: Relative standard deviations <20% at LOQ levels
Matrix Effects and Quality Control
Even with effective SPE cleanup, some matrix effects may persist. Matrix-matched calibration or standard addition methods are recommended for accurate quantification. The use of isotopically labeled internal standards further improves method accuracy and precision by correcting for extraction efficiency variations and matrix effects.
Quality control measures should include analysis of method blanks, spiked samples, and certified reference materials when available. For routine analysis, the inclusion of control samples at low, medium, and high concentration levels ensures ongoing method performance verification.
Advanced Applications and Future Directions
Recent advancements in SPE technology include the development of PRiME HLB cartridges that eliminate conditioning and equilibration steps, saving processing time and solvent costs. These next-generation sorbents maintain excellent cleanup efficiency while simplifying workflow.
For high-throughput applications, 96-well SPE plates offer automated processing capabilities. Poseidon Scientific’s 96-well SPE plates provide consistent performance for large sample batches, making them ideal for regulatory testing laboratories and quality control facilities.
Looking forward, the integration of online SPE with LC-MS/MS systems offers promising opportunities for automated, high-throughput mycotoxin analysis. These systems combine extraction, concentration, and analysis in a single workflow, potentially revolutionizing routine mycotoxin testing in grain commodities.
Technical Note: SPE Cartridge Selection
For mycotoxin applications in grain matrices, Poseidon Scientific recommends HLB cartridges in appropriate sizes based on sample load and expected interference levels. The HLB SPE cartridges are available in various configurations (1 cc/30 mg to 35 cc/6 g) to accommodate different sample volumes and matrix complexities. For particularly challenging matrices containing high lipid content, consider using larger sorbent masses or specialized cleanup approaches.