1. Common Antibiotics Monitored in Dairy Testing
Dairy product safety requires rigorous monitoring of antibiotic residues, with beta-lactams and tetracyclines being the most frequently analyzed classes. Beta-lactams, including penicillins (amoxicillin, ampicillin) and cephalosporins, are widely used in veterinary medicine for treating mastitis and other infections. Tetracyclines such as oxytetracycline, tetracycline, and chlortetracycline are broad-spectrum antibiotics commonly administered to dairy cattle.
Regulatory bodies like the European Union and FDA establish maximum residue limits (MRLs) for these compounds, typically ranging from 4-100 μg/kg depending on the specific antibiotic and dairy product. The challenge in monitoring these residues lies in their diverse chemical properties—beta-lactams are generally polar and labile, while tetracyclines can form complexes with metal ions and exhibit zwitterionic behavior.
Recent studies have demonstrated that mixed-mode SPE cartridges containing both hydrophobic and cation exchange functionalities provide superior recovery for these diverse antibiotic classes compared to single-mode sorbents. The ability to retain compounds through multiple interaction mechanisms makes mixed-mode SPE particularly valuable for multi-residue antibiotic analysis in complex matrices like milk.
2. Matrix Challenges in Milk
Milk presents one of the most challenging matrices for analytical chemistry due to its complex composition. The primary interferences include:
- Proteins (3-4%): Casein and whey proteins can bind to antibiotics, particularly tetracyclines, reducing extraction efficiency and causing matrix effects in LC-MS/MS analysis.
- Fats (3-5%): Lipophilic components can interfere with extraction and cause column fouling in chromatographic systems.
- Lactose (4-5%): While water-soluble, high concentrations can affect solvent polarity and extraction efficiency.
- Minerals: Calcium and magnesium ions can complex with tetracyclines, affecting their recovery and stability.
Research from Simpson (2000) highlights that milk samples exist as suspensions with moderate viscosity, requiring specialized dispersion techniques for effective sample preparation. The high protein and lipid content often leads to emulsification during liquid-liquid extraction, making SPE a more reliable alternative for consistent recovery and cleaner extracts.
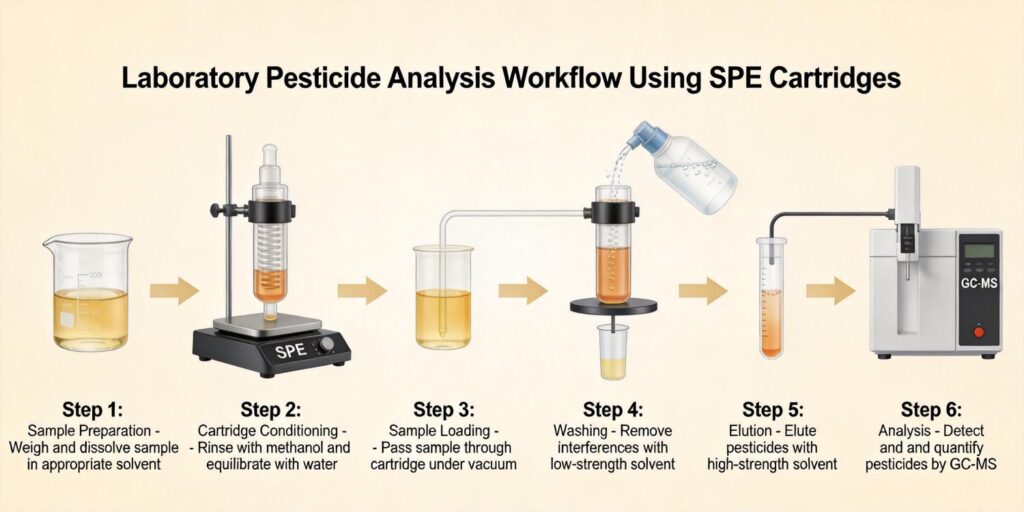
3. Sample Pretreatment Steps Before SPE
Effective sample pretreatment is crucial for successful SPE of antibiotic residues from milk. The standard protocol includes:
- Protein Precipitation: Addition of acids (trichloroacetic acid, metaphosphoric acid) or organic solvents (acetonitrile, methanol) to denature and precipitate proteins. A common approach uses metaphosphoric acid/methanol (3:2 v/v) for effective deproteinization.
- Defatting: Centrifugation or filtration to remove lipid components after protein precipitation.
- pH Adjustment: Critical for ionizable antibiotics. For tetracyclines, EDTA-containing buffers (pH 4.0) help prevent metal complexation. For beta-lactams, slightly acidic conditions (pH 4-5) improve stability.
- Dilution: Typically 1:1 dilution with appropriate buffer to reduce matrix effects and optimize loading conditions.
Van Poucke et al. (1991) demonstrated that blending 5-10 mL of milk with C18 material in a syringe barrel eliminated the need for mortar/pestle blending and reduced sample loss. This approach allows for larger sample volumes, enhancing method sensitivity for trace-level antibiotic detection.
4. Cartridge Selection Strategies
HLB vs Mixed-Mode Cartridges
The choice between hydrophilic-lipophilic balanced (HLB) and mixed-mode cartridges depends on the target analytes and required selectivity:
HLB Cartridges
Poseidon Scientific’s HLB SPE cartridges feature a water-wettable polymer that retains compounds through reversed-phase mechanisms. Advantages include:
- High capacity for polar compounds
- Compatibility with pH 0-14 solvents
- No requirement for activation/equilibration in some formats
- Effective for broad-spectrum antibiotic screening
However, HLB cartridges may provide less selective cleanup compared to mixed-mode options, potentially requiring additional purification steps.
Mixed-Mode Cartridges
For antibiotic residue analysis, mixed-mode cartridges offer superior selectivity. Poseidon Scientific provides several options:
- MCX cartridges: Combine reversed-phase and strong cation exchange for basic compounds (pKa ≥ 4.5)
- MAX cartridges: Combine reversed-phase and strong anion exchange for acidic compounds (pKa 2-8)
- WAX cartridges: Weak anion exchange for strong acids (pKa < 1)
- WCX cartridges: Weak cation exchange for strong bases (pKa > 10)
Simpson and Van Horne (1993) demonstrated that mixed-mode cartridges containing alkyl chains and cation exchange groups provide optimal recovery for substances with acidic, neutral, or basic properties. For tetracyclines (which exhibit zwitterionic behavior) and beta-lactams (often polar and ionizable), mixed-mode MCX or MAX cartridges typically yield cleaner extracts with higher recovery rates.
5. Optimized Washing and Elution Conditions
Proper washing and elution are critical for removing interferences while maintaining high antibiotic recovery:
Washing Steps
For mixed-mode cartridges (MCX/MAX):
- Initial wash: 2-5% methanol in water to remove polar interferences
- Secondary wash: Acidic or basic solutions to disrupt ionic interactions with non-target compounds
- Optional tertiary wash: Organic solvents to remove hydrophobic interferences
For HLB cartridges: Typically use 5% methanol in water for washing, effectively removing salts and polar matrix components while retaining antibiotics.
Elution Optimization
Elution conditions must disrupt both hydrophobic and ionic interactions:
- MCX cartridges: 5% ammonium hydroxide in methanol effectively elutes basic antibiotics by neutralizing cation exchange interactions
- MAX cartridges: 2% formic acid in methanol for acidic antibiotics
- HLB cartridges: 90:10 acetonitrile:methanol or methanol with 0.1% formic acid
Studies show that allowing cartridges to soak with elution solvent for 0.5-1 minute before collection improves recovery by 10-15%. Multiple small-volume elutions (e.g., 2 × 1.5 mL) often yield better recovery than single large-volume elutions.
6. LC-MS/MS Detection Parameters
Liquid chromatography coupled with tandem mass spectrometry provides the sensitivity and selectivity required for antibiotic residue analysis at regulatory limits:
Chromatographic Conditions
- Column: C18 or phenyl-hexyl columns (100 × 2.1 mm, 1.7-2.6 μm)
- Mobile phase: 0.1% formic acid in water (A) and 0.1% formic acid in acetonitrile (B)
- Gradient: Typically 5-95% B over 10-15 minutes
- Flow rate: 0.2-0.4 mL/min
- Temperature: 30-40°C
MS/MS Parameters
- Ionization: Electrospray ionization (ESI) in positive mode for most antibiotics
- Source temperature: 150-300°C
- Desolvation gas: 600-1000 L/hr
- Cone gas: 50-150 L/hr
- Collision energy: Compound-specific optimization (typically 10-40 eV)
Multiple reaction monitoring (MRM) using two transitions per compound provides both quantification and confirmation, meeting EU and FDA guidelines for residue analysis.
7. Recovery Validation and Regulatory Considerations
Recovery Validation
Method validation for antibiotic residue analysis must demonstrate:
- Recovery: 70-120% across the calibration range
- Precision: ≤15% RSD for intermediate precision
- Linearity: R² ≥ 0.99 over the working range
- Limit of quantification: At or below regulatory MRLs
- Specificity: No interference from matrix components
Studies using mixed-mode SPE cartridges have demonstrated recoveries of 85-98% for antibiotics in milk with RSDs of 2-8%, meeting validation criteria for regulatory compliance.
Regulatory Guidelines
European Union Regulations:
- Commission Regulation (EU) No 37/2010 establishes MRLs for veterinary medicinal products
- Decision 2002/657/EC outlines performance criteria for analytical methods
- Requires identification points system for confirmatory methods
FDA Guidelines:
- Title 21 CFR Part 556 establishes tolerances for residues
- FDA Compliance Program 7371.004 covers milk safety
- Requires validation according to FDA Guidance for Industry #118
Both regulatory frameworks emphasize the importance of robust sample preparation methods. SPE, particularly using mixed-mode cartridges, has become the gold standard for antibiotic residue analysis due to its ability to provide clean extracts, high recovery, and compatibility with sensitive detection techniques like LC-MS/MS.
Implementation Considerations
For laboratories implementing antibiotic residue analysis in milk:
- Start with mixed-mode cartridges for better selectivity, especially when analyzing multiple antibiotic classes
- Consider 96-well SPE plates for high-throughput applications—Poseidon Scientific’s 96-well SPE plates offer automation compatibility
- Validate with incurred samples in addition to spiked samples to account for matrix-binding effects
- Implement quality controls at multiple levels to ensure method robustness
The combination of optimized SPE workflows with sensitive LC-MS/MS detection provides laboratories with reliable tools for ensuring dairy product safety and regulatory compliance in today’s stringent food safety environment.