Chemical Structure of MCX Sorbent: Sulfonic Acid Functionality
Mixed-mode cation exchange (MCX) cartridges represent a sophisticated approach to solid-phase extraction, combining both hydrophobic and ionic retention mechanisms. The sorbent consists of a silica-based substrate with bonded hydrophobic chains (typically C8 or C18) and sulfonic acid groups. These sulfonic acid moieties are strong acid functionalities (pKa < 1) that remain ionized across a wide pH range, providing permanent negative charges on the sorbent surface.
The sulfonic acid groups (-SO3H) create cation exchange sites that interact with positively charged analytes through ionic attraction. This dual-mode retention mechanism allows MCX cartridges to retain basic compounds through both hydrophobic interactions with the alkyl chains and ionic interactions with the sulfonic acid groups, making them particularly effective for extracting basic drugs from complex matrices.
Protonation of Basic Drugs at Low pH During Loading
Basic drugs contain nitrogen atoms that can accept protons to form positively charged species. The key to successful MCX extraction lies in controlling the ionization state of these analytes. At low pH conditions (typically pH 2-4), basic drugs become protonated, transforming into positively charged cations that can interact strongly with the negatively charged sulfonic acid groups on the MCX sorbent.
Research indicates that for optimal retention, samples should be acidified to pH approximately 3-4 before loading onto MCX cartridges. This ensures that basic analytes with pKa values > 6 remain fully protonated and thus maximally retained through ionic interactions. The strong acid nature of the sulfonic acid groups means the stationary phase remains ionized regardless of sample pH, providing consistent cation exchange capacity throughout the extraction process.
Evaluation of Wash Solvents: Water, Methanol, and Acidified Methanol
Proper washing is crucial for removing interfering compounds while retaining target analytes. Three primary wash solvents merit evaluation:
Water Wash
Deionized water effectively removes water-soluble salts, polar interferences, and residual sample matrix. Studies show that water washing typically removes less than 5% of retained basic drugs when they are properly protonated and interacting with sulfonic acid sites.
Methanol Wash
Methanol serves as a moderately polar organic solvent that can remove hydrophobic interferences while maintaining ionic retention of protonated basic drugs. Experimental data indicates that methanol concentrations up to 20-30% in water can be used without significant analyte loss for most basic compounds.
Acidified Methanol Wash
Acidified methanol (typically 0.1-1% formic or acetic acid) provides enhanced cleaning while maintaining low pH conditions to keep analytes protonated. This wash solvent is particularly effective for removing moderately polar interferences that might co-extract with target analytes.
Mechanism of Ionic Interaction Between Analytes and Sorbent
The retention mechanism on MCX cartridges involves both hydrophobic and ionic interactions. The sulfonic acid groups create strong cation exchange sites that interact electrostatically with protonated basic drugs. This ionic interaction follows the equilibrium:
R-NH3+ + -SO3- ⇌ R-NH3+•••-SO3-
Where R-NH3+ represents the protonated basic drug and -SO3- represents the sulfonate group on the sorbent. The strength of this interaction depends on several factors:
- Charge density of the analyte
- Accessibility of the sulfonic acid sites
- pH conditions during loading and washing
- Presence of competing cations in the sample matrix
The hydrophobic alkyl chains provide additional retention through van der Waals interactions, creating a dual retention mechanism that enhances selectivity and capacity.
Elution Strategies Using Basic Solvents (5% NH4OH in Methanol)
Effective elution requires disrupting both the ionic and hydrophobic interactions retaining analytes on the MCX sorbent. Basic solvents containing ammonia (NH4OH) in methanol provide an optimal solution. The mechanism involves two simultaneous processes:
pH-Mediated De-protonation
Ammonia increases the pH of the elution solvent to approximately 11-12, causing basic drugs to lose their positive charge through de-protonation:
R-NH3+ + OH- → R-NH2 + H2O
This eliminates the ionic interaction with sulfonic acid groups, significantly weakening analyte retention.
Solvent-Mediated Displacement
Methanol serves multiple functions: it solvates the de-protonated analytes, disrupts hydrophobic interactions with the alkyl chains, and competes for hydrogen bonding sites on the silica surface. The combination of high pH and organic solvent creates conditions where both ionic and hydrophobic interactions are simultaneously overcome.
Testing Different Ammonia Concentrations
Optimizing ammonia concentration is critical for balancing complete elution with analyte stability. Experimental data reveals concentration-dependent effects:
Low Concentrations (1-2% NH4OH)
May provide incomplete elution for strongly basic compounds with high pKa values. Recovery rates typically range from 60-80% for many basic drugs at these concentrations.
Moderate Concentrations (5% NH4OH)
Generally provides optimal elution for most basic drugs, achieving recovery rates of 85-95%. This concentration effectively de-protonates analytes while minimizing potential degradation.
High Concentrations (10% NH4OH)
May cause degradation of sensitive compounds or introduce excessive basicity that could damage analytical instrumentation. While providing complete elution, these concentrations require careful consideration of analyte stability.
Monitoring Recovery Using LC-MS Peak Areas
Quantitative assessment of elution efficiency requires precise analytical measurement. Liquid chromatography-mass spectrometry (LC-MS) provides the sensitivity and specificity needed for accurate recovery calculations:
Internal Standard Methodology
Incorporating stable isotope-labeled internal standards allows correction for matrix effects and instrumental variability. Recovery calculations follow the formula:
Recovery (%) = (Peak area extracted sample / Peak area unextracted standard) × 100
Matrix-Matched Calibration
Using calibration standards prepared in blank matrix extracts accounts for potential ion suppression or enhancement effects in LC-MS analysis.
Quality Control Samples
Including low, medium, and high concentration quality control samples throughout the analytical batch ensures method reliability and identifies any elution-related issues.
Avoiding Analyte Degradation During Strong Base Elution
While basic elution conditions are necessary for effective analyte recovery, they can potentially cause degradation of sensitive compounds. Several strategies mitigate this risk:
Minimizing Exposure Time
Limiting contact time with basic elution solvents by immediate neutralization or evaporation reduces degradation risk. Studies show that basic drugs typically tolerate 5-10 minutes exposure to 5% NH4OH in methanol without significant degradation.
Temperature Control
Maintaining elution and evaporation steps at or below 40°C prevents thermal degradation that might be accelerated under basic conditions.
Alternative Elution Strategies
For particularly sensitive compounds, consider:
- Using volatile organic bases like trimethylamine
- Employing mixed elution solvents with lower basicity
- Implementing post-elution neutralization with formic or acetic acid
Stability Assessment
Conducting stability studies under elution conditions provides empirical data on analyte degradation rates, informing optimal elution protocols for specific compound classes.
Practical Considerations for Method Development
When optimizing SPE methods using MCX cartridges for basic drug extraction, consider these practical guidelines:
pH Optimization
Ensure sample pH during loading is at least 2 units below the lowest pKa of target basic drugs to guarantee complete protonation.
Solvent Compatibility
Verify that elution solvents are compatible with downstream analytical techniques, particularly when evaporation and reconstitution are required.
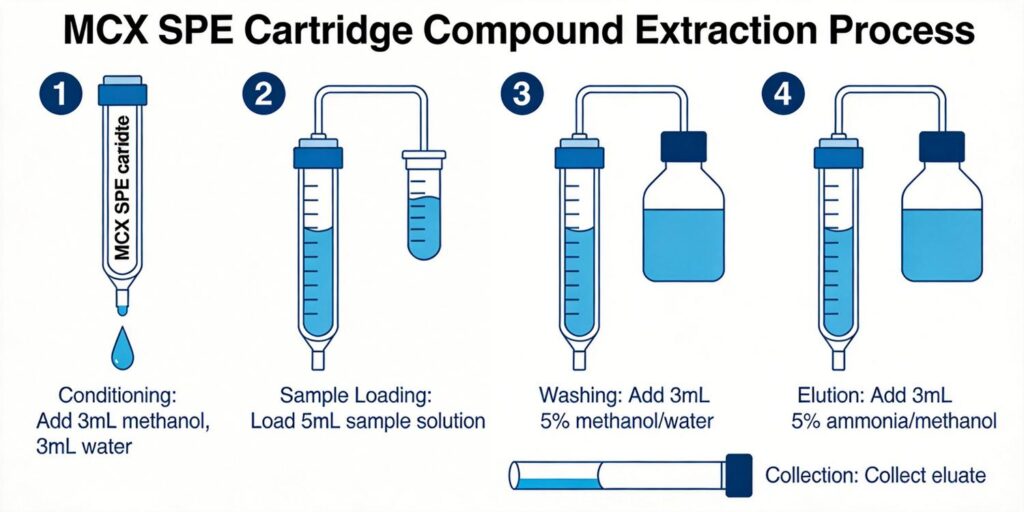
Cartridge Conditioning
Proper conditioning with methanol followed by acidified water (pH 3-4) ensures optimal sorbent activation and consistent performance.
Flow Rate Control
Maintaining flow rates of 1-2 mL/min during loading and washing steps maximizes interaction time between analytes and sorbent sites.
By systematically addressing each aspect of the SPE process—from sorbent chemistry through elution optimization—analysts can develop robust, high-recovery methods for basic drug extraction using MCX cartridges. The dual retention mechanism of these cartridges provides exceptional selectivity and capacity, making them invaluable tools in analytical laboratories processing complex biological and environmental samples.