Major Plasma Interferences: Phospholipids, Proteins, and Salts
Plasma presents one of the most challenging matrices for analytical chemists due to its complex composition. The three primary interference categories that significantly impact LC-MS analysis are phospholipids, proteins, and salts. Phospholipids are particularly problematic as they cause matrix effects, ion suppression, shorten column lifetime, increase MS maintenance costs, and elevate LC-MS quantitative variability. According to Waters documentation, phospholipids are “the main cause of matrix effects, ion suppression, shortened column life, increased MS maintenance costs, and increased LC-MS quantitative variability.”
Proteins present another major challenge, with plasma containing approximately 60-80 g/L of total protein. These macromolecules can clog analytical columns, bind to analytes (reducing recovery), and interfere with detection. Salts, including sodium, potassium, chloride, and bicarbonate ions, can cause ion suppression in MS detection and interfere with chromatographic separation. The combination of these interferences necessitates sophisticated sample preparation strategies to achieve reliable analytical results.
MCX Mixed-Mode Retention Principle
Mixed-mode cation exchange (MCX) solid-phase extraction represents a powerful approach for plasma sample cleanup. MCX sorbents combine two distinct retention mechanisms: reversed-phase (hydrophobic) interactions and strong cation exchange (SCX) functionality. This dual-mode approach provides orthogonal selectivity that surpasses single-mode SPE materials.
The MCX sorbent features sulfonic acid groups (SO₃⁻) that provide strong cation exchange capacity of approximately 1 meq/g. These groups selectively retain protonated basic compounds through ionic interactions. Simultaneously, the hydrophobic polymer backbone provides reversed-phase retention for neutral and acidic compounds. This combination allows for selective retention of basic analytes (pKa ≥ 4.5) while effectively removing phospholipids and other matrix interferences.
The water-wettable polymeric structure of MCX sorbents remains stable across the entire pH range (0-14), enabling flexible method development. Unlike silica-based sorbents, MCX materials contain no silanol groups that could complicate retention mechanisms or method development.
Plasma Pretreatment Options: Protein Precipitation vs Dilution
Before MCX SPE, plasma samples typically require pretreatment to reduce protein content and viscosity. Two primary approaches exist: protein precipitation and dilution.
Protein Precipitation: This method involves adding organic solvents (typically acetonitrile or methanol) or acids to denature and precipitate proteins. While effective for protein removal, precipitation can co-precipitate analytes and may not adequately address phospholipid interference. According to literature, “Proteins may be precipitated if necessary. Protein binding of isolates may be a problem and should be considered if recoveries of isolate standards are high but recoveries from sample are low.”
Dilution: Simple dilution with aqueous buffers or water represents a gentler approach. For MCX applications, dilution with acidic buffers (typically containing 4% H₃PO₄) serves multiple purposes: it reduces protein concentration, maintains analytes in protonated form for optimal cation exchange retention, and provides appropriate ionic strength. Waters recommends diluting plasma samples 1:1 (v/v) with solutions containing 4% H₃PO₄ in 200 mM ammonium formate, resulting in final concentrations of 100 mM ammonium formate and 2% H₃PO₄.
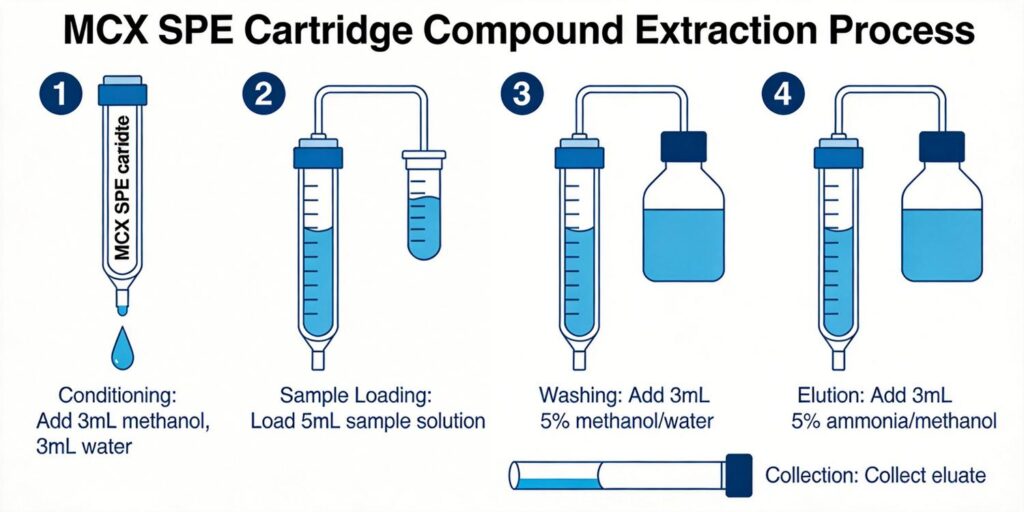
Conditioning and Equilibration Steps
Traditional SPE protocols require conditioning with organic solvent followed by equilibration with aqueous solution to activate the sorbent. However, modern MCX sorbents, particularly water-wettable polymeric versions, often eliminate these steps. The Oasis PRiME MCX technology, for instance, requires “no conditioning/equilibration” steps, significantly simplifying the workflow.
When using standard MCX cartridges, a typical conditioning sequence includes:
- 2 mL methanol (or other suitable organic solvent)
- 2 mL water or appropriate buffer
These steps ensure the sorbent is properly solvated and ready to receive the aqueous sample. The conditioning solvent should match or be miscible with the sample solvent to prevent precipitation at the sorbent interface.
Sample Loading pH Optimization
pH control during sample loading represents the most critical parameter for successful MCX extraction. Basic analytes must be protonated to interact effectively with the cation exchange sites. The optimal loading pH should be at least 2 pH units below the pKa of the target basic compounds.
For comprehensive screening of basic drugs, literature recommends starting at pH 2.2: “The starting pH of 2.2 results in less ionization of the acidic drugs and hence, better retention on the cartridge.” This acidic environment ensures that basic analytes with pKa values as low as approximately 4.2 remain protonated and retained.
For specific applications, loading pH can be optimized between pH 2-4. Acidic buffers containing formic acid, phosphoric acid, or ammonium formate/formic acid mixtures provide both pH control and appropriate ionic strength. The buffer concentration should be sufficient to maintain pH but not so high as to compete with analyte retention.
Wash Sequence Design to Remove Phospholipids
Effective phospholipid removal represents a key advantage of MCX SPE. Phospholipids, being zwitterionic or anionic at acidic pH, exhibit minimal retention on MCX sorbents and can be efficiently washed away. A well-designed wash sequence typically includes:
- Initial aqueous wash: 2% formic acid in water or similar acidic solution removes salts, polar interferences, and unretained phospholipids.
- Organic wash: Methanol or methanol/water mixtures (typically 20-100% methanol) remove neutral lipids and remaining phospholipids while maintaining basic analytes in protonated, retained form.
- Optional second wash: For particularly challenging matrices, an additional wash with 5% ammonium hydroxide or other basic organic solutions can remove acidic interferences.
Waters’ Oasis PRiME MCX protocols demonstrate effective phospholipid removal: “More simple: clearer method can remove up to 99% of phospholipids, which are the main cause of matrix effects, ion suppression, shortened column life, increased MS maintenance costs, and increased LC-MS quantitative variability.”
Strong Base Elution Solvent Preparation
Elution of basic analytes from MCX sorbents requires disruption of both ionic and hydrophobic interactions. This is typically achieved using basic organic solvents that deprotonate the analytes, neutralizing their positive charge and weakening cation exchange interactions.
The standard elution solvent consists of 5% ammonium hydroxide in methanol. Alternative basic eluents include:
- 2-10% ammonium hydroxide in methanol
- 2% ammonium hydroxide in acetonitrile/methanol mixtures
- Ammoniated ethyl acetate (2% NH₄OH)
- Ammoniated methylene chloride/isopropanol mixtures
Preparation should ensure complete mixing and stability. Ammonium hydroxide solutions should be prepared fresh or verified for concentration stability. The elution volume typically ranges from 1-3 mL, depending on cartridge size and analyte characteristics. Complete elution often requires two aliquots of elution solvent.
Validation: Recovery and Matrix Effect Evaluation
Comprehensive validation of MCX SPE methods requires assessment of both recovery and matrix effects. Recovery evaluation involves comparing analyte responses in extracted samples versus neat standards at equivalent concentrations. Acceptable recoveries typically range from 70-120%, though specific applications may have different requirements.
Matrix effects represent a more critical parameter for LC-MS applications. These are evaluated using post-extraction spiking experiments:
- Prepare blank plasma extracts using the MCX method
- Spike known concentrations of analytes into the extracts
- Compare responses with neat standards at equivalent concentrations
- Calculate matrix factor (MF) = Response in matrix/Response in neat solution
Matrix effects ≤ 20% (MF 0.8-1.2) are generally acceptable. MCX SPE typically provides excellent matrix effect reduction, with Waters reporting “removes up to 99% of phospholipids” and corresponding reduction in ion suppression.
Additional validation parameters should include:
- Precision (intra-day and inter-day)
- Accuracy
- Linearity across the calibration range
- Limit of detection and quantification
- Carryover assessment
- Stability under processing conditions
The orthogonal selectivity of MCX SPE, combining reversed-phase and cation exchange mechanisms, provides superior cleanup compared to single-mode approaches. As noted in literature, “mixed-mode cartridge providing hydrophobic and cation exchange interactions, combined with a pH-dependent sample application and extraction, can give high recoveries of analytes from plasma… and the resulting SPE eluates are easily amenable to subsequent GC- and HPLC-analysis.”
For laboratories implementing MCX SPE for plasma analysis, the combination of proper pH control during loading, optimized wash sequences, and appropriate elution conditions delivers clean extracts with minimal matrix effects, enabling reliable quantification even at trace levels.