Principles of Ion Exchange Chemistry
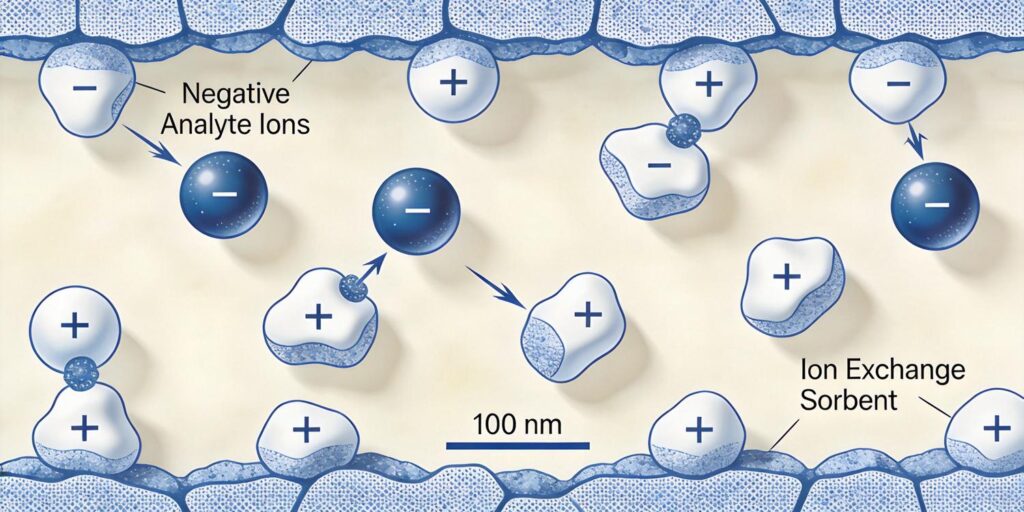
Ion exchange solid phase extraction (SPE) represents one of the most selective and powerful sample preparation techniques available to analytical chemists. Unlike traditional reversed-phase SPE that relies on hydrophobic interactions, ion exchange SPE utilizes electrostatic interactions between charged analytes and oppositely charged functional groups on the sorbent surface. This fundamental difference provides exceptional selectivity for ionic compounds that might otherwise be difficult to isolate from complex matrices.
The core principle of ion exchange SPE involves the reversible exchange of ions between a solid sorbent and a liquid sample. When a charged analyte encounters an oppositely charged functional group on the sorbent, electrostatic attraction occurs, forming what is essentially an ionic bond. This interaction is strong enough to retain analytes while allowing neutral and similarly charged interferences to pass through during washing steps.
As noted in forensic applications literature, “Ionic interactions occur between charged sorbent and analyte of opposite charge. The pH is manipulated to ionize analytes functional group. Ionic bonds are strong and retain analytes. Hydrophobic interferences can be washed away with organic solvents. Polar interferences can be removed with aqueous or weak aqueous/organic washes.” This dual-wash capability makes ion exchange SPE particularly effective for complex sample matrices.
The kinetics of ion exchange differ significantly from other SPE mechanisms. Research indicates that “the kinetics of ion exchange are slower than those of non-polar or polar mechanisms. Several causes could be cited but the most important are the slow diffusion of ionic species (often because they also drag with them a sheath of water molecules, resulting in a diffusion radius that is much larger than the ion itself) and the process of displacement of the counter-ion that is associated with the ion exchanger.” This has practical implications for method development, particularly regarding flow rates and contact times.
Strong vs Weak Ion Exchange
Understanding the distinction between strong and weak ion exchangers is crucial for successful method development. The fundamental difference lies in their pH-dependent behavior and charge characteristics.
Strong Ion Exchangers
Strong ion exchangers maintain their charge across the entire pH range. Common examples include:
- Strong Cation Exchangers (SCX): Benzenesulfonic acid (pKa <1) and propylsulfonic acid (pKa <1)
- Strong Anion Exchangers (SAX): Quaternary amine (always charged)
As explained in forensic SPE literature, “Strong ion-exchangers are charged across the entire pH range. Therefore, the only neutralization that can be done to release a compound is the neutralization of the charge on the compound itself. When you put a strong acid or base on a strong anion or cation exchanger you have no way to break the ionic bond since both sorbent and analyte are always charged.” This characteristic makes strong ion exchangers ideal for weak acids and bases where pH manipulation can effectively control analyte ionization.
Weak Ion Exchangers
Weak ion exchangers exhibit pH-dependent ionization and are only charged within specific pH ranges:
- Weak Cation Exchangers (WCX): Carboxylic acid (pKa ~4.8)
- Weak Anion Exchangers (WAX): Primary amine (pKa ~9.8), secondary amine (pKa ~10.1-10.9), diethylamino (pKa ~10.6)
The literature clarifies that “weak ion-exchangers are only charged in a given pH range and can be neutralized to release strong anions and cations. When using weak ion-exchangers with weak acids or bases, we can choose to neutralize either the compounds themselves or the packing depending on what conditions we hope to achieve in the final elution.” This flexibility makes weak ion exchangers particularly valuable for extracting strong acids and bases.
Cation vs Anion Exchange
Cation Exchange Extractions
Cation exchange sorbents carry negative charges and are designed to retain positively charged analytes. According to forensic applications data, “Cation-exchange sorbents are negatively charged while basic analytes are manipulated to carry a positive charge. Opposites attract, forming strong bonds.”
Common cation exchange sorbents include:
- Benzenesulfonic acid (strong)
- Propylsulfonic acid (strong)
- Carboxylic acid (weak)
Typical applications for cation exchange include extraction of basic drugs, catecholamines, pharmaceuticals, herbicides, and metal ions. These extractions are normally performed from aqueous matrices, with basic elution solvents used to neutralize the analyte.
Anion Exchange Extractions
Anion exchange sorbents carry positive charges and retain negatively charged analytes. The literature states that “Anion-exchange sorbents are positively charged, whereas acidic analytes are manipulated to carry negative charges. Again, opposites attract forming strong bonds.”
Common anion exchange sorbents include:
- Primary amine (weak)
- Aminopropyl (weak)
- Diethylamino (weak)
- Quaternary amine (strong)
Typical analytes extracted using anion exchange include phosphates, acidic drugs, organic acids, vitamins, and similar compounds. These extractions are normally performed from aqueous matrices, with acidic elutions used to neutralize the analytes of interest.
How pH Affects Retention
pH control represents the single most critical parameter in ion exchange SPE. The relationship between pH and analyte retention follows well-established principles based on acid-base chemistry and pKa values.
The pH-pKa Relationship
For effective ion exchange retention, both the sorbent and analyte must be appropriately charged. The general rule states that to ensure 99% or more ionization, the pH should be at least two pH units below the pKa of a cation and two pH units above the pKa of an anion. This principle is summarized in forensic literature: “The number of molecules with charged cationic groups increases at pH values below the molecules pKa value. The number of molecules with charged anionic groups decreases at pH values below the molecule’s pKa value.”
A practical summary table from forensic applications illustrates this relationship:
| Functionality | State | 2 < pKa | 1 < pKa | At pKa | 1 < pKa | 2 < pKa |
|---|---|---|---|---|---|---|
| Acid | Anionic (-) | 1 | 5 | 50 | 91 | 99 |
| Base | Cationic (+) | 99 | 91 | 50 | 5 | 1 |
Practical pH Considerations
For cation exchange extractions, the goal during loading is to promote bonding by maintaining pH < analyte pKa or pH > sorbent pKa. Elution occurs by disrupting bonding through pH > analyte pKa or pH < sorbent pKa.
For anion exchange extractions, loading requires pH > analyte pKa or pH < sorbent pKa, while elution requires pH < analyte pKa or pH > sorbent pKa.
It’s important to note that quoted pKa values pertain to specific conditions of temperature, concentration, and environment. As noted in the literature, “The effective acidity or basicity of a functional group close to a bonded silica surface may be very different. Consequently, you may use quoted values of molecular properties as a guide for selection of experimental conditions of pH, but it is always wise to verify the selection by experiment.”
Counter-Ion Selectivity
pH isn’t the only factor affecting retention and elution. Counter-ion selectivity plays a crucial role in ion exchange processes. Different ionic species show varying affinities for ion exchange sorbents, expressed as relative selectivities. Two general trends are observed:
- Selectivity increases as the charge of the ion increases (divalent ions are generally more retentive than monovalent ones)
- Aqueous solubility and selectivity vary approximately inversely
For example, fluoride ion (F–) represents a low selectivity ion due to its good solvation, while iodide (I–) exhibits high selectivity due to poor solvation.
Typical Analytical Applications
Pharmaceutical and Clinical Applications
Ion exchange SPE finds extensive application in pharmaceutical analysis and clinical toxicology. Mixed-mode sorbents combining ion exchange with reversed-phase functionality have revolutionized drug analysis. As described in the literature, “The cartridge is first conditioned in the normal manner with methanol and water, and then the analytes are applied under acidic conditions (pH approximately 6). The basic analytes (pKa > 6) will be ionized and retained by ionic interactions. The acids, although partially ionized, should be retained along with the neutral drugs by hydrophobic, reversed-phase interactions.”
This approach allows for class separation of acidic, neutral, and basic compounds from a single sample aliquot, significantly improving analytical efficiency.
Forensic Toxicology
In forensic applications, ion exchange SPE enables comprehensive drug screening from biological matrices. The literature describes how “copolymeric bonded phases” can be used to separate drugs into their respective classes. These materials “have found a lot of use in drug abuse applications, where wide ranges of components need to be extracted in a single procedure. Selective elution allows for the removal of interferences.”
Typical forensic applications include:
- Basic drug analysis using cation exchange
- Acidic drug analysis using anion exchange
- Comprehensive screening using mixed-mode sorbents
- Metal ion analysis in environmental and biological samples
Environmental Analysis
Ion exchange SPE proves invaluable for environmental applications, particularly for:
- Herbicide and pesticide analysis
- Organic acid monitoring in water samples
- Metal speciation studies
- Pharmaceutical residue analysis in wastewater
Food and Beverage Analysis
The food industry utilizes ion exchange SPE for:
- Organic acid profiling in beverages
- Mycotoxin analysis
- Vitamin analysis in fortified products
- Additive and preservative monitoring
Method Development Considerations
When developing ion exchange SPE methods, several practical considerations emerge from the literature:
- Sample Preparation: “Ion exchange functions most reliably when the samples are aqueous… It is common to apply a dilute buffer to the sample prior to performing ion exchange extraction. The buffer serves two purposes: 1) Control the pH of the sample and hence the ionic state of the analyte and potential interferences. 2) Modify the ionic strength of the sample.”
- Flow Rates: “Sample loading rates are more commonly specified when ion exchange is the mode of extraction and it is not easy to elute analytes in very small volumes.”
- Capacity Considerations: “When using ion-exchangers, capacity is not determined purely by the organic loading of the phase, although it is related to it. When determining how much compound an ion-exchanger will hold, you need to know what its milliequivalence is. This is a calculation of the charge-to-mass ratio of the packing material.”
Modern Developments
Recent advancements in ion exchange SPE include specialized sorbents like Oasis MAX (Mixed-mode Anion eXchange) for acidic compounds and Oasis WCX (Weak Cation eXchange) for strong basic compounds. These materials offer tightly controlled ion-exchange capacities and improved reproducibility for challenging analytes.
For laboratories working with diverse analyte types, sorbent selection kits containing MCX, MAX, WCX, and WAX sorbents enable rapid method development and optimization for unknown analytes, zwitterionic compounds, or mixtures with different retention/elution properties.
Understanding ion exchange principles in SPE cartridges provides analytical chemists with powerful tools for selective sample preparation. By mastering the relationships between pH, pKa, sorbent selection, and application requirements, laboratories can develop robust methods that deliver clean extracts and reliable results across diverse analytical challenges.