Key Principles of SPE Method Robustness
When designing SPE methods for LC-MS applications, robustness is not merely a desirable feature—it’s an absolute necessity. A robust SPE method, as defined in the literature, should yield near-quantitative recovery over the entire desired concentration range with acceptable precision both within and between laboratories, and across different time periods. This is particularly critical for LC-MS applications where matrix effects can significantly impact ionization efficiency and detection sensitivity.
The fundamental principles of robust SPE method design include:
- Consistent Recovery: SPE recoveries should exceed 90% absolute recovery. Values below this threshold typically indicate issues with solubility, pH adjustment, or solvent strength optimization.
- Matrix Tolerance: The method must handle natural variations in sample matrices, whether biological fluids, environmental samples, or pharmaceutical formulations.
- Reproducibility: Both intra-day and inter-day precision should meet analytical requirements, typically with RSD values below 15% for bioanalytical methods.
- Transferability: Methods should perform consistently across different laboratories, operators, and equipment.
As noted in SPE literature, “SPE is but one step in the analytical method, which normally consists of sample pretreatment, extraction, elution and analysis. Attention paid to the design of the SPE method should certainly be no less than that of the analytical method itself.”
Sorbent Chemistry Selection Criteria
Choosing the appropriate sorbent chemistry is the cornerstone of successful SPE method development. The selection process should consider both analyte characteristics and matrix properties:
Analyte Characterization
- Structure and Functional Groups: Identify polar, non-polar, and ionizable groups
- pKa Values: Determine ionization states at different pH values
- Solubility Profile: Understand solubility in aqueous and organic solvents
- Stability Considerations: Assess stability under various pH and solvent conditions
Matrix Characterization
- pH and Ionic Strength: Natural variations in sample matrices
- Interference Profile: Similar functional groups, pKa values, and polarity
- Protein Content: Critical for biological samples to prevent MS source contamination
- Qualitative and Quantitative Variability: Natural variations in sample composition
For LC-MS applications, mixed-mode sorbents combining hydrophobic and ion-exchange interactions often provide superior selectivity. As demonstrated in forensic applications, copolymeric phases have shown significant advantages over traditional C18 or C8 phases for extracting multiple drug classes from complex biological matrices.
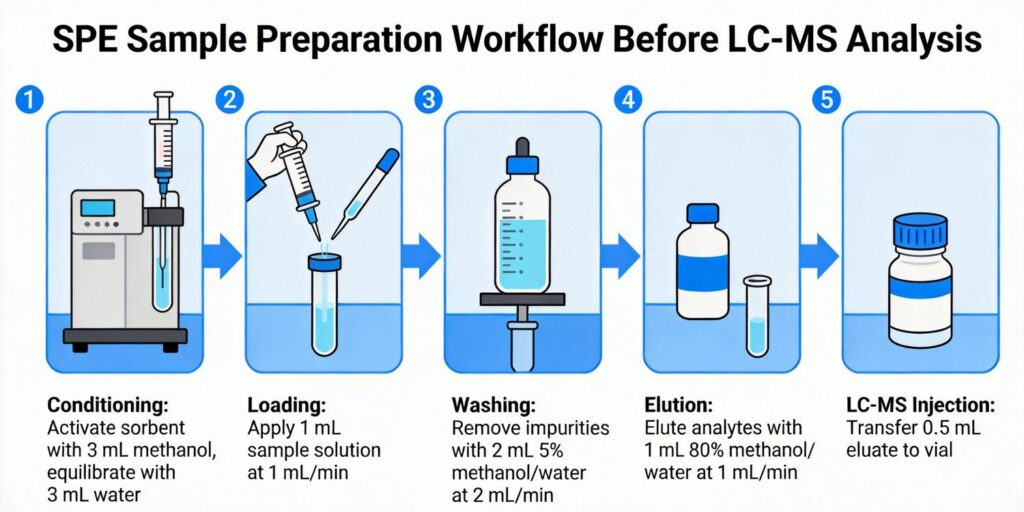
Optimizing Conditioning and Equilibration Steps
Proper conditioning and equilibration are critical for ensuring consistent sorbent performance and reproducible recoveries. These steps prepare the sorbent bed to accept the sample in the appropriate solvent environment.
Conditioning Protocol
- Strong Solvent Wash: Typically methanol or acetonitrile to wet the sorbent and remove any contaminants
- Weak Solvent Equilibration: Water or buffer solution matching the sample loading conditions
- Flow Rate Control: Maintain consistent flow rates (typically 1-3 drops/second) to ensure proper solvent exchange
Critical Considerations
- Solvent Compatibility: Ensure the conditioning solvent is stronger than the elution solvent when using reversed-phase sorbents
- Bed Dryness Prevention: Never allow the sorbent bed to dry between conditioning and sample loading
- Buffer Matching: For ion-exchange sorbents, the conditioning buffer should match the sample pH and ionic strength
Recent advancements include non-conditioned SPE technologies that simplify protocols while maintaining performance, particularly valuable for high-throughput LC-MS applications.
Balancing Washing Strength vs Analyte Retention
The washing step represents a critical optimization point where maximum matrix removal must be balanced against analyte retention. This balance directly impacts both recovery and extract cleanliness.
Wash Solvent Optimization Strategy
- Identify Maximum Retention Conditions: Determine which sorbents provide maximum analyte retention
- Evaluate Elution Profiles: Identify which eluent solvents yield highest recoveries
- Optimize Wash Strength: Determine the strongest wash solvent that will not elute the analyte
- Assess Cleanliness: Evaluate eluate cleanliness under conditions of maximum analyte retention
Practical Approaches
- Stepwise Wash Optimization: Begin with weak wash solvents and gradually increase strength
- pH Adjustment: For ionizable compounds, adjust wash pH to maximize retention of target analytes while removing interferences
- Organic Content Optimization: For reversed-phase SPE, optimize organic solvent percentage in wash solutions
- Ionic Strength Control: For ion-exchange SPE, optimize salt concentration in wash buffers
For LC-MS applications, washing with pure water after appropriate buffer washes can eliminate excess ions that might interfere with MS ionization processes, improving system performance.
Elution Solvent Optimization
Elution optimization focuses on achieving complete analyte recovery in the smallest possible volume while maintaining compatibility with downstream LC-MS analysis.
Elution Strategy Development
- Solvent Strength Selection: Choose solvents strong enough to disrupt analyte-sorbent interactions
- Volume Optimization: Determine minimum volume for complete elution
- Compatibility Assessment: Ensure elution solvent is compatible with LC-MS analysis
- Multiple Elution Consideration: For complex samples, consider sequential elution with different solvents
LC-MS Specific Considerations
- Ion Suppression Minimization: Use pure organic solvents without modifiers or buffer ions when possible
- Source Compatibility: Ensure elution solvents don’t cause source contamination or ionization suppression
- Concentration Factor: Optimize for maximum concentration while maintaining solubility
- Evaporation Compatibility: Consider solvent volatility if evaporation and reconstitution are required
For mixed-mode sorbents, elution often requires disruption of both hydrophobic and ionic interactions, typically achieved through pH adjustment combined with organic solvent.
Method Validation Experiments
Comprehensive validation ensures the SPE method meets analytical requirements and regulatory standards. Key validation parameters include:
Critical Variables to Validate
- Sorbent Performance: Different cartridges, lots, and sorbent weights
- Conditioning Parameters: Strong and weak solvent volumes and compositions
- Loading Conditions: Organic percentage, pH, ionic strength, and volume
- Wash Optimization: Organic percentage, pH, ionic strength, and volume
- Elution Parameters: Volume, organic percentage, and composition
- Flow Rate Effects: Loading, wash, and elution flow rates
Performance Assessment
- Linearity and Range: Different analyte concentrations and matrix loadings
- Recovery Determination: Absolute and relative recovery across concentration range
- Precision Evaluation: Intra-day and inter-day precision
- Matrix Effect Assessment: Ion suppression/enhancement in LC-MS
- Stability Studies: Analyte stability in loading solvent and eluent
As emphasized in SPE literature, “The analyst will look for 100% recoveries, confident that quantitative extraction and elution has occurred in this case. Values greater than 100% may indicate contamination of the sample or extract from a source other than the sample.”
Documentation and SOP Development
Proper documentation and Standard Operating Procedure (SOP) development ensure method transferability, regulatory compliance, and long-term robustness.
SOP Essential Components
- Detailed Procedure: Step-by-step instructions with precise volumes, times, and conditions
- Equipment Specifications: Specific equipment models, settings, and maintenance requirements
- Reagent Specifications: Grade, purity, preparation methods, and storage conditions
- Quality Control Procedures: Acceptance criteria, control samples, and troubleshooting guidelines
- Safety Considerations: Handling precautions, waste disposal, and emergency procedures
Documentation Best Practices
- Batch Records: Complete documentation of each extraction batch
- Equipment Logs: Maintenance, calibration, and performance verification records
- Training Records: Operator competency assessment and training documentation
- Change Control: Documentation of any method modifications and re-validation
- Data Integrity: Complete audit trail of all method development and validation data
Automation Considerations
For automated SPE systems, documentation should include:
- Method Programming: Complete method parameters and programming details
- System Verification: Regular performance verification procedures
- Carryover Assessment: Documentation of carryover testing and mitigation strategies
- Throughput Optimization: Documentation of throughput optimization experiments
As noted in automation literature, “Successful introduction of automation requires the laboratory to realize that an automated SPE workstation is a tool to help the laboratory personnel perform their jobs, but is not a replacement for them.”
Conclusion
Designing robust SPE methods for LC-MS applications requires systematic approach combining fundamental chromatographic principles with practical optimization strategies. By carefully considering sorbent selection, optimizing each SPE step, conducting comprehensive validation, and developing thorough documentation, analysts can create methods that deliver consistent, reliable performance even in demanding high-throughput environments.
The integration of SPE with LC-MS has revolutionized analytical capabilities, enabling sensitive detection at picogram levels while maintaining high throughput. As SPE technology continues to evolve with new sorbent chemistries, formats, and automation capabilities, the principles of robust method design remain essential for ensuring analytical success.
For laboratories implementing SPE methods, Poseidon Scientific offers a comprehensive range of SPE products including HLB SPE cartridges, MAX SPE cartridges, MCX SPE cartridges, WAX SPE cartridges, WCX SPE cartridges, and 96-well SPE plates designed to meet the demanding requirements of modern LC-MS applications.