1. Veterinary Drugs Commonly Monitored in Food Products
The analysis of veterinary drug residues in food products represents a critical component of food safety monitoring programs worldwide. These compounds span multiple therapeutic classes, each presenting unique analytical challenges for LC-MS detection. The most commonly monitored veterinary drugs include:
Antibacterial Agents
Sulfonamides (sulfamethazine, sulfadimethoxine), tetracyclines (oxytetracycline, chlortetracycline), beta-lactams (penicillins, cephalosporins), and macrolides (erythromycin, tylosin) constitute the primary antibacterial classes requiring monitoring. These compounds exhibit diverse chemical properties, ranging from acidic sulfonamides to amphoteric tetracyclines, necessitating tailored SPE approaches.
Antiparasitic Compounds
Ivermectin, doramectin, and other avermectins represent important antiparasitic agents with extremely low maximum residue limits (MRLs) in edible tissues. Their high lipophilicity and low concentration requirements demand exceptional extraction efficiency and clean-up protocols.
Growth Promoters and Hormones
Anabolic steroids, corticosteroids, and beta-agonists (clenbuterol, ractopamine) require specialized extraction strategies due to their potent biological activity at trace levels and complex metabolic pathways.
Other Therapeutic Classes
Anthelmintics, coccidiostats, and tranquilizers complete the spectrum of veterinary drugs monitored in food safety programs. Each class presents unique challenges in terms of polarity, stability, and matrix interference.
According to Simpson and Wynne (2000), “The versatility and wide acceptance of SPE in this field is such that a chapter specifically on this subject is provided later in this book.” This underscores the critical role SPE plays in veterinary drug analysis across diverse food matrices.
2. Matrix Challenges in Meat and Milk Samples
Food matrices, particularly meat and milk, present formidable challenges for veterinary drug residue analysis. These complex biological materials contain numerous interfering compounds that can compromise LC-MS/MS detection if not properly addressed during sample preparation.
Protein and Lipid Content
Meat samples typically contain 15-25% protein and varying lipid content depending on tissue type and animal species. Milk contains approximately 3.5% protein and 3.5-5% fat. These macromolecules can bind analytes, reduce extraction efficiency, and cause matrix effects during ionization. As noted in the literature, “Protein binding of isolates may be a problem and should be considered if recoveries of isolate standards are high but recoveries from sample are low.”
Endogenous Compounds
Meat and milk contain numerous endogenous compounds including peptides, amino acids, carbohydrates, vitamins, and minerals that can interfere with analysis. The high protein and lipid content of these samples often makes them prone to emulsification during liquid-liquid extraction, making SPE particularly valuable for such applications.
Sample Viscosity and Heterogeneity
As described in veterinary applications, “Horse urine and urine collected from other grazing animals is comparatively viscous and may in some cases present extreme difficulties in processing through SPE cartridges.” While this specifically addresses urine, similar viscosity challenges can occur with homogenized tissue samples and milk.
Species-Specific Variations
Different animal species exhibit variations in matrix composition that affect extraction efficiency. For instance, equine blood has a relatively high-packed cell volume compared to other species, resulting in lower plasma yields for extraction.
The literature emphasizes that “identifying and eliminating interferences and troubleshooting low recoveries requires good knowledge of the matrix composition.” This principle is particularly crucial for meat and milk samples where matrix effects can significantly impact method performance.
3. Sample Extraction Strategies Prior to SPE
Effective sample extraction before SPE is essential for releasing bound analytes and preparing the matrix for clean-up. The choice of extraction strategy depends on the analyte properties, matrix type, and required sensitivity.
Homogenization and Protein Precipitation
For solid tissues, mechanical homogenization followed by solvent extraction represents the standard approach. A typical method involves homogenizing tissue with aqueous methanol or acetonitrile, often acidified to improve analyte recovery. As demonstrated by Horie et al. (1991), “antibacterial drugs were extracted from fish tissue by homogenizing in a slurrying buffer of metaphosphoric acid/methanol (3:2 v/v) to deproteinize the sample.”
Liquid-Liquid Extraction (LLE)
While SPE has largely replaced LLE for many applications, certain scenarios still benefit from preliminary LLE. For fatty samples, initial extraction with non-polar solvents followed by back-extraction into aqueous phase can effectively remove lipids before SPE clean-up.
Matrix Solid Phase Dispersion (MSPD)
MSPD represents an innovative approach that combines sample disruption and extraction in a single step. As described in the literature, “MSPD is a low-tech process that can be applied as easily in the field as at the laboratory bench. It achieves disruption and extraction of solid or viscous samples.” For milk analysis, Van Poucke et al. (1991) demonstrated that “5.0 to 10.0 mL of milk blended with 2.0 g of C18 material” could effectively extract sulfonamide residues with approximately 95% recovery.
Enzymatic Digestion
For certain analytes bound to proteins or trapped in cellular structures, enzymatic digestion with proteases or lipases can improve extraction efficiency. This approach is particularly valuable for conjugated metabolites or protein-bound residues.
pH Adjustment and Buffer Systems
Strategic pH adjustment during extraction can optimize analyte solubility and minimize protein binding. Buffer systems like phosphate, acetate, or TRIS buffers help maintain consistent extraction conditions and improve reproducibility.
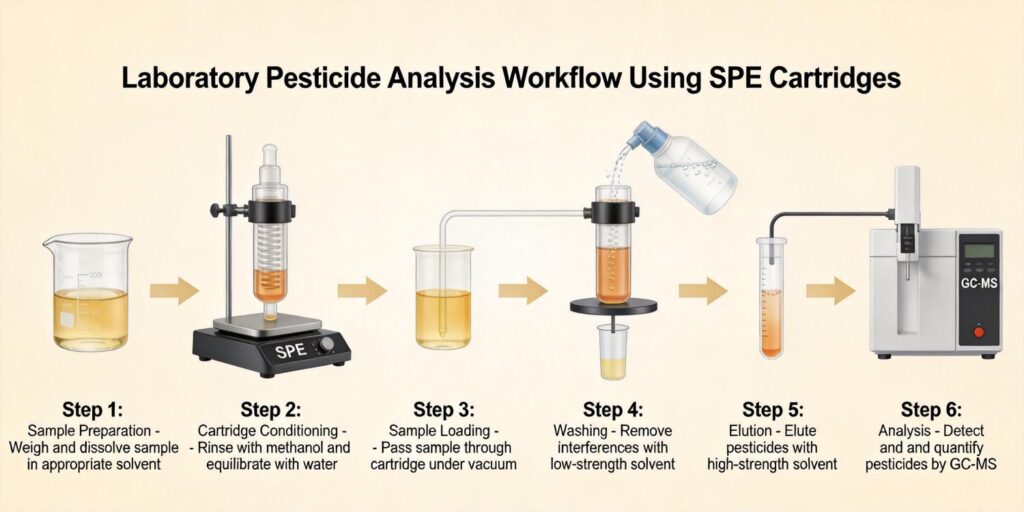
The fundamental SPE strategy, as outlined in analytical literature, “comprises the isolation (and concentration) of the analytes from a complex matrix by adsorption onto an appropriate sorbent, the removal of interfering impurities by washing with a suitable solvent system and then the selective recovery of the retained analytes.” Proper pre-extraction ensures this process proceeds efficiently.
4. Sorbent Selection for Veterinary Drug Classes
Selecting the appropriate SPE sorbent is critical for achieving optimal recovery and clean-up for veterinary drug analysis. The choice depends on analyte properties, matrix composition, and analytical requirements.
Reversed-Phase Sorbents (C18, C8, HLB)
Hydrophilic-lipophilic balanced (HLB) sorbents like those offered by Poseidon Scientific provide excellent retention for a wide range of veterinary drugs. These water-wettable polymers maintain retention even when run dry, making them ideal for complex biological matrices. For lipophilic compounds like avermectins, traditional C18 sorbents offer strong retention but may require careful conditioning to prevent channeling.
Mixed-Mode Sorbents (MCX, MAX, WAX, WCX)
Mixed-mode sorbents combine reversed-phase and ion-exchange mechanisms for enhanced selectivity. MCX (Mixed-mode Cation Exchange) sorbents are ideal for basic compounds like beta-agonists and certain antibiotics. MAX (Mixed-mode Anion Exchange) sorbents effectively capture acidic compounds such as sulfonamides and tetracyclines. For zwitterionic compounds, WAX (Weak Anion Exchange) and WCX (Weak Cation Exchange) sorbents offer pH-dependent retention capabilities.
Specialized Sorbents for Specific Applications
For particular veterinary drug classes, specialized sorbents may offer advantages. Florisil (magnesium silicate) effectively removes lipids and pigments from extracts. Graphitized carbon black shows exceptional retention for planar molecules and can remove chlorophyll and other plant pigments in feed analysis.
Sorbent Capacity and Particle Size Considerations
The literature emphasizes that “bed mass has upon recovery and extract purity” in veterinary drug applications. For meat and milk samples with high interference loads, larger bed masses (200-500 mg) typically provide better clean-up, though they may require larger elution volumes. Smaller particle sizes (30-50 μm) offer improved kinetics and efficiency but may require reduced flow rates.
The decision tree approach to sorbent selection, as illustrated in analytical texts, considers analyte polarity, ionizability, and molecular weight. However, as Simpson and Wynne (2000) note, “This approach fails to take into account a potentially bigger variable: intersample matrix variations,” emphasizing that matrix considerations often override theoretical predictions in veterinary drug analysis.
5. Washing Protocols for Lipid Removal
Effective washing protocols are essential for removing lipids and other matrix interferences while retaining target analytes. The high lipid content of meat and milk samples necessitates carefully optimized washing strategies.
Water-Based Washes
Initial washing with 5-10% methanol or acetonitrile in water effectively removes polar interferences while retaining most veterinary drugs on reversed-phase sorbents. For ion-exchange sorbents, washing with appropriate buffer solutions at optimized pH removes neutral and oppositely charged interferences.
Organic Solvent Washes for Lipid Removal
For lipid-rich samples, washing with hexane, heptane, or methyl tert-butyl ether (MTBE) effectively removes non-polar lipids without eluting most veterinary drugs. As demonstrated in analytical methods, “the column was washed with two 1-ml portions of n-hexane-dichloromethane (7:3, v/v)” to remove lipid interferences while retaining target analytes.
Acidified or Basified Washes
For ionizable compounds, washing with acidified or basified organic solvents can selectively remove interferences with similar hydrophobicity but different ionization states. For example, washing MCX cartridges with acidified methanol removes neutral lipids while retaining basic analytes in their protonated form.
Sequential Washing Strategies
Multi-step washing protocols often provide superior clean-up. A typical sequence might include: (1) water or dilute buffer to remove salts and polar interferences, (2) water-miscible organic solvent (5-20% methanol) to remove moderately polar interferences, and (3) water-immiscible organic solvent to remove lipids.
Washing Volume Optimization
Washing volumes must balance clean-up efficiency with analyte retention. Excessive washing can lead to analyte breakthrough, particularly for moderately retained compounds. Method development should include washing volume optimization studies to establish the maximum volume that maintains acceptable recovery (>85%).
The literature on biological sample preparation emphasizes that “optimizing the washing procedure” is a critical step in method development. For veterinary drug analysis in food matrices, washing optimization directly impacts method sensitivity by reducing matrix effects during LC-MS/MS analysis.
6. Elution Solvent Optimization
Elution solvent selection and optimization determine final extract purity, analyte recovery, and compatibility with LC-MS analysis. Veterinary drugs span a wide polarity range, necessitating carefully tailored elution conditions.
Solvent Strength and Selectivity
For reversed-phase sorbents, methanol, acetonitrile, and acetone provide varying elution strengths and selectivities. Methanol generally offers stronger elution for polar compounds, while acetonitrile provides better elution for less polar analytes. As noted in method descriptions, analytes are typically “eluted with two 1.5-ml portions of methanol” following appropriate washing.
Acidified or Basified Eluents
For ion-exchange mechanisms, elution requires disrupting ionic interactions through pH adjustment or competing ion addition. Acidic eluents (formic acid, acetic acid in organic solvent) effectively elute basic compounds from cation exchangers, while basic eluents (ammonia, ammonium hydroxide in organic solvent) elute acidic compounds from anion exchangers.
Mixed-Mode Elution Strategies
For mixed-mode sorbents, elution typically requires both organic solvent to disrupt hydrophobic interactions and pH adjustment or ion competition to disrupt ionic interactions. A common approach uses methanol or acetonitrile containing 2-5% formic acid or ammonium hydroxide.
Elution Volume Optimization
Minimizing elution volume improves concentration factors and detection limits. Elution profile studies determine the minimum volume required for complete analyte recovery. Typically, 2-3 bed volumes of elution solvent provide >95% recovery for most veterinary drugs.
Solvent Evaporation and Reconstitution
Following elution, solvent evaporation and reconstitution in mobile phase-compatible solvents prepare extracts for LC-MS analysis. Careful evaporation conditions (temperature, gas flow) prevent analyte loss or degradation. Reconstitution in initial mobile phase composition minimizes chromatographic issues.
The literature emphasizes that “optimizing the elution procedure” is essential for achieving both high recovery and clean extracts. For veterinary drug analysis, elution optimization must consider not only recovery but also extract cleanliness to minimize matrix effects in LC-MS/MS.
7. LC-MS/MS Detection and Validation
LC-MS/MS represents the gold standard for veterinary drug residue analysis due to its exceptional sensitivity, selectivity, and capability for multi-residue analysis. Proper SPE sample preparation directly impacts LC-MS/MS performance and method validation outcomes.
Chromatographic Considerations
Reversed-phase chromatography with C18 or C8 columns remains standard for veterinary drug analysis. Mobile phase additives like formic acid, ammonium formate, or ammonium acetate enhance ionization and improve peak shape. Gradient elution efficiently separates diverse veterinary drug classes in single runs.
Mass Spectrometric Detection
Triple quadrupole instruments operating in multiple reaction monitoring (MRM) mode provide the sensitivity and selectivity required for veterinary drug analysis at regulatory limits. Electrospray ionization (ESI) accommodates most veterinary drugs, though atmospheric pressure chemical ionization (APCI) may benefit certain non-polar compounds.
Matrix Effects Assessment
Matrix effects represent a critical validation parameter for LC-MS/MS methods. Post-extraction addition, post-column infusion, and echo-peak techniques assess ionization suppression or enhancement. Effective SPE clean-up minimizes matrix effects, improving method accuracy and precision.
Method Validation Parameters
Complete method validation for veterinary drug analysis includes assessment of: selectivity/specificity, linearity, accuracy (recovery), precision (repeatability, intermediate precision), limit of detection (LOD), limit of quantification (LOQ), matrix effects, and stability. Regulatory guidelines from organizations like FDA, EU, and Codex Alimentarius provide specific requirements.
Quality Control and Assurance
Implementation of quality control samples (blanks, fortified samples, continuing calibration checks) ensures ongoing method performance. Proficiency testing and participation in inter-laboratory comparisons validate method accuracy against peer laboratories.
High-Throughput Applications
For laboratories processing large sample volumes, 96-well SPE plates offer significant throughput advantages. Automated systems can process entire plates simultaneously, improving productivity and reproducibility while reducing analyst time.
The integration of optimized SPE protocols with sensitive LC-MS/MS detection enables reliable quantification of veterinary drug residues at levels far below maximum residue limits. As the literature notes, “Modern drug candidates are often very potent substances, for which reason the doses administered in preclinical and clinical studies are relatively low. Thus, assay sensitivity is a major goal.” This principle applies equally to veterinary drug residue analysis, where detection at part-per-billion or part-per-trillion levels is often required.
In conclusion, successful SPE sample preparation for LC-MS analysis of veterinary drugs in meat and milk requires careful consideration of analyte properties, matrix challenges, and analytical requirements. By implementing optimized extraction, sorbent selection, washing, and elution protocols, analysts can achieve the sensitivity, selectivity, and reliability demanded by modern food safety monitoring programs.