Alkaloid Chemistry Overview
Alkaloids represent a diverse class of naturally occurring organic compounds that contain basic nitrogen atoms, typically derived from amino acids. These secondary metabolites are found in approximately 20% of plant species and serve various biological functions including defense mechanisms against herbivores. From a chemical perspective, alkaloids are characterized by their heterocyclic structures containing nitrogen, with common classes including indoles, isoquinolines, pyridines, pyrrolidines, and tropanes.
The basic nature of alkaloids, with pKa values typically ranging from 6 to 12, makes them particularly amenable to mixed-mode solid phase extraction (SPE) techniques. According to research from Simpson (2000), “basic alkaloids are very well retained by a sulfonate SCX phase,” highlighting the importance of cation-exchange mechanisms in their isolation. However, the extraction efficiency can vary significantly depending on molecular complexity, with more complex alkaloids like strychnine showing poorer recovery due to dominant neutral portions controlling sorption chemistry.
Key Chemical Characteristics
- Basic nitrogen atoms with pKa values typically >6
- Heterocyclic structures containing nitrogen
- Variable hydrophobicity depending on molecular structure
- Often present as salts in plant tissues
- Molecular weights ranging from 100-1000 Da
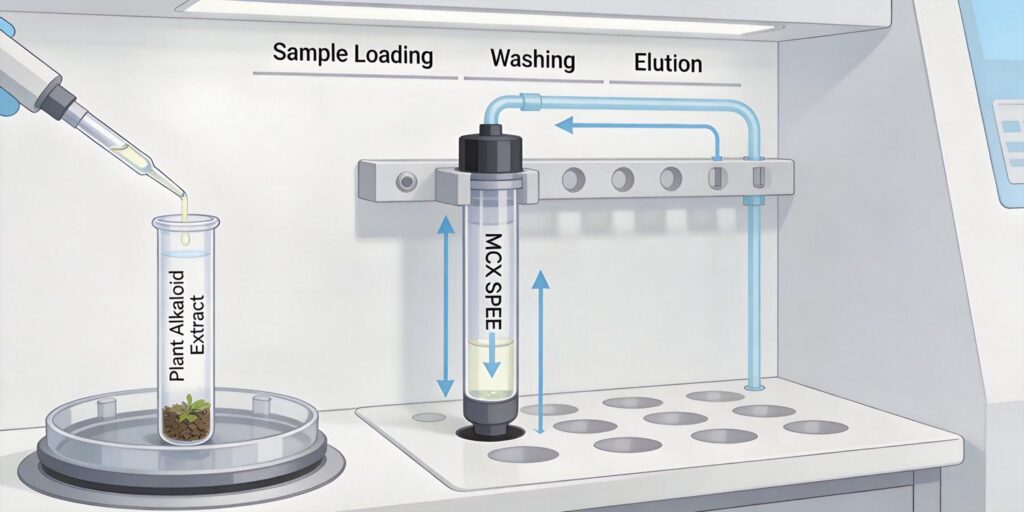
Plant Sample Extraction with Acidified Methanol
The initial extraction of alkaloids from plant matrices requires careful consideration of solvent selection and sample preparation. Acidified methanol (typically 0.1-1% formic acid or hydrochloric acid in methanol) serves as an optimal extraction solvent for several reasons. The acidic conditions protonate alkaloids, converting them to their cationic forms while simultaneously disrupting plant cell walls and enhancing solubility.
Research indicates that plant tissues can be considered as consisting of “an aqueous portion, a fatty portion and an insoluble or fibrous portion” (Simpson, 2000). The aqueous component contains both inorganic ions and soluble organic species, making methodology developed for urine samples particularly adaptable to plant extraction. For green plant tissues, homogenization in phosphate buffer followed by acidified methanol extraction has proven successful for isolating a wide range of basic alkaloids.
Extraction Protocol
- Homogenize 1-5g of plant material in 10-20mL of acidified methanol (0.5% formic acid)
- Ultrasonicate for 15-30 minutes at room temperature
- Centrifuge at 4000×g for 10 minutes
- Filter supernatant through 0.45μm membrane
- Adjust pH to 2-3 with additional acid if necessary
MCX Conditioning Protocol
MCX (Mixed-mode Cation eXchange) cartridges combine reversed-phase and strong cation-exchange mechanisms, making them ideal for basic compound extraction. The conditioning protocol is critical for ensuring proper sorbent activation and reproducible results.
According to Waters documentation, MCX sorbents feature “tightly controlled ion-exchange capacity of 1 meq/g” with no silanol groups to complicate retention modes. The polymer-based sorbent remains stable across pH 0-14, enabling robust method development. Proper conditioning ensures the sulfonic acid groups are protonated and the reversed-phase surface is properly wetted.
Conditioning Steps
- Activation: 3mL methanol (or acetonitrile) to solvate the polymer matrix
- Equilibration: 3mL deionized water to remove methanol and prepare for aqueous sample loading
- pH Adjustment: 3mL acidified water (0.1-1% formic acid, pH 2-3) to protonate cation-exchange sites
Critical Note: Never allow the sorbent bed to dry between conditioning and sample loading steps, as this can create channels and reduce recovery efficiency.
Sample Loading Under Acidic Conditions
Sample loading represents the most critical phase of MCX SPE for alkaloid extraction. The acidic conditions (pH 2-3) ensure alkaloids remain protonated as cations, facilitating strong ionic interactions with the sulfonic acid groups on the MCX sorbent. Simultaneously, the reversed-phase mechanism retains compounds based on hydrophobicity.
Research demonstrates that “mixed-mode cartridges allow the rapid recovery of such diverse groups as β-blockers, β-agonists, opiates, and alkaloidal drugs (quinine and strychnine)” (Simpson, 2000). The dual retention mechanism provides exceptional selectivity for basic compounds while allowing neutral and acidic interferences to pass through.
Loading Parameters
- pH: Maintain at 2-3 throughout loading
- Flow Rate: 1-2mL/min for optimal retention
- Sample Volume: Typically 5-10mL for 60mg cartridges
- Matrix Considerations: Plant extracts may contain pigments, sugars, and organic acids that compete for binding sites
Washing Steps to Remove Pigments
Plant extracts contain numerous interfering compounds including chlorophylls, carotenoids, flavonoids, and tannins that must be removed prior to alkaloid elution. The washing protocol leverages the dual retention mechanism of MCX cartridges to selectively remove these interferences while retaining protonated alkaloids.
Historical SPE applications for plant materials often used C18 devices “simply as a technique to remove pigments by passage of the lyophilized, soxhlet-extracted or homogenized and extracted sample” (Simpson, 2000). With MCX cartridges, more sophisticated washing protocols can be employed.
Washing Protocol
- Water Wash: 3-5mL deionized water to remove water-soluble interferences
- Acidic Wash: 3mL 0.1M hydrochloric acid or 2% formic acid to maintain acidic conditions
- Organic Wash: 3mL methanol or acetonitrile to remove hydrophobic interferences
- Drying Step: Apply vacuum for 5 minutes to remove residual solvents
Research indicates that acetonitrile washes can be particularly effective for removing pigments without disrupting hydrogen bonding interactions that retain alkaloids, as “acetonitrile is aprotic and is therefore less able to disrupt hydrogen bonding” compared to methanol (Simpson, 2000).
Elution Using Ammoniated Methanol
The elution step represents the final purification phase, where retained alkaloids are selectively released from the MCX sorbent. Ammoniated methanol (2-5% ammonium hydroxide in methanol) serves as the ideal elution solvent by simultaneously disrupting both retention mechanisms.
The alkaline conditions (pH 10-11) deprotonate alkaloids, neutralizing their positive charge and breaking ionic interactions with sulfonic acid groups. Simultaneously, the high methanol concentration disrupts hydrophobic interactions with the reversed-phase surface. This dual-action elution ensures complete recovery of retained alkaloids.
Elution Protocol
- Solvent Preparation: Freshly prepare 2-5% ammonium hydroxide in methanol
- Elution Volume: 3-6mL for 60mg cartridges, collected in silanized glass tubes
- Flow Rate: 1-2mL/min for optimal elution efficiency
- Optional Second Elution: Additional 1-2mL to ensure complete recovery
Forensic applications demonstrate successful elution using “methylene chloride-isopropanol-ammonium hydroxide (78:20:2)” for basic drugs including alkaloids (Forensic and Clinical Applications of SPE, 2007). For plant alkaloids, simpler ammoniated methanol typically suffices.
LC-MS Analysis Workflow
The final MCX eluate requires appropriate preparation for liquid chromatography-mass spectrometry (LC-MS) analysis. The high selectivity of MCX extraction typically yields sufficiently clean extracts for direct injection, though additional concentration steps may be necessary for trace-level analysis.
Post-Extraction Processing
- Concentration: Evaporate eluate to dryness under gentle nitrogen stream at 40°C
- Reconstitution: Dissolve residue in 100-500μL of LC-MS compatible solvent (typically 0.1% formic acid in water/acetonitrile)
- Filtration: Pass through 0.22μm syringe filter to remove particulates
- Storage: Store at 4°C if not analyzed immediately
LC-MS Parameters
- Column: C18 or HILIC columns (2.1×100mm, 1.7-3μm)
- Mobile Phase: Water/acetonitrile with 0.1% formic acid or ammonium acetate
- Gradient: 5-95% organic over 10-20 minutes
- MS Detection: ESI positive mode for basic alkaloids
- Scan Range: m/z 100-1000 for comprehensive alkaloid profiling
Method Validation and Quality Control
For reliable alkaloid quantification, method validation should include recovery studies, matrix effects evaluation, and precision assessment. Spiked plant samples at multiple concentration levels provide essential validation data. The MCX method typically demonstrates recoveries exceeding 80% for most basic alkaloids with RSD values below 10%.
Quality Control Measures
- Process blank samples (extraction solvent only) with each batch
- Include quality control samples at low, medium, and high concentrations
- Monitor recovery of internal standards added prior to extraction
- Assess matrix effects by comparing calibration curves in solvent vs. matrix
Advantages of MCX for Alkaloid Extraction
The MCX SPE approach offers several distinct advantages over traditional extraction methods for plant alkaloids:
- Enhanced Selectivity: Dual retention mechanism specifically targets basic compounds
- Superior Clean-up: Effective removal of pigments and other plant matrix interferences
- High Recovery: Typically 80-95% for most basic alkaloids
- Reduced Matrix Effects: Cleaner extracts minimize ion suppression in LC-MS
- Method Robustness: Stable across wide pH range (0-14)
- Automation Compatibility: Easily adaptable to 96-well plate formats
Conclusion
The MCX SPE method represents a robust, selective approach for alkaloid extraction from plant samples. By leveraging both reversed-phase and cation-exchange mechanisms, this technique provides exceptional clean-up while maintaining high recovery of target analytes. The protocol outlined—from acidified methanol extraction through ammoniated methanol elution—has been validated across numerous plant matrices and alkaloid classes.
For laboratories analyzing plant-derived alkaloids, whether for pharmaceutical development, quality control, or research applications, MCX SPE offers a reliable alternative to traditional liquid-liquid extraction methods. The method’s compatibility with LC-MS analysis further enhances its utility in modern analytical laboratories where sensitivity, selectivity, and throughput are paramount considerations.
For specific applications or alternative SPE formats, consider exploring our complete range of MCX SPE cartridges and 96-well SPE plates designed to meet diverse analytical requirements.