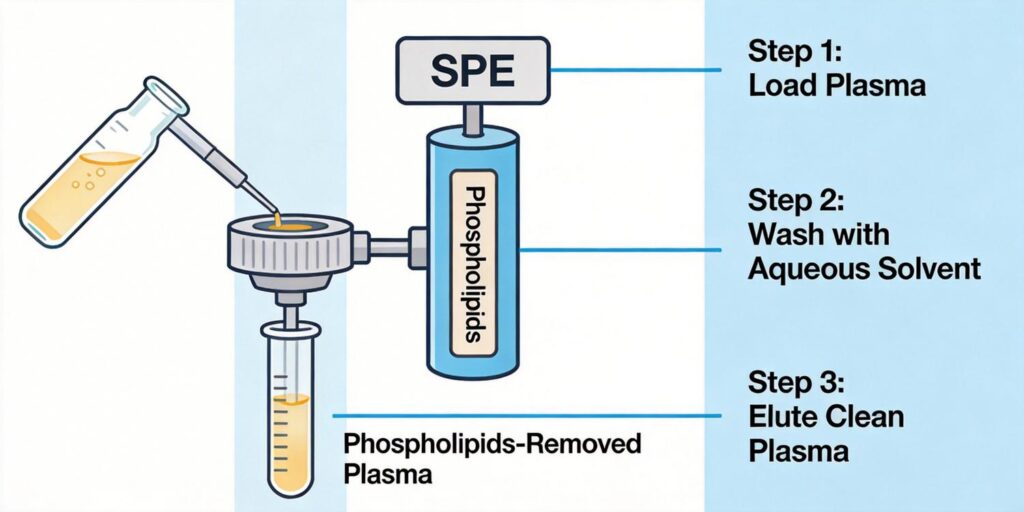
Introduction: The Phospholipid Challenge in Plasma Analysis
Phospholipids represent one of the most persistent and problematic interferences in plasma analysis, particularly when using LC-MS detection. These endogenous compounds not only cause matrix effects through ion suppression but also contribute to signal variability, reduced column lifetime, and increased instrument maintenance costs. As Dr. Xu, product manager at Poseidon Scientific, I’ve seen firsthand how proper SPE methodology can transform analytical outcomes by effectively removing these troublesome compounds.
Step 1: Identifying Phospholipid Interference Ions
Common Phospholipid m/z Signatures
Before developing any SPE method, it’s crucial to understand the specific phospholipid ions that cause interference. In LC-MS analysis, phospholipids typically appear as characteristic ions in the m/z range of 400-900. The most problematic phospholipids include:
- Phosphatidylcholines (PC): m/z 496.3, 520.3, 524.3, 758.5, 786.5
- Lysophosphatidylcholines (LPC): m/z 496.3, 520.3, 524.3
- Phosphatidylethanolamines (PE): m/z 716.5, 744.5, 768.5
- Sphingomyelins (SM): m/z 703.6, 731.6
Monitoring Strategies
During method development, monitor these characteristic ions in both blank plasma extracts and spiked samples. As noted in SPE literature, “matrix components can have a dramatic influence on sorbent-analyte and analyte-sample interactions” (Simpson, 2000). This is particularly true for phospholipids, which can compete with analytes for binding sites and cause unpredictable recovery variations.
Step 2: Selecting Optimal Sorbent Chemistry
Mixed-Mode Sorbents for Comprehensive Removal
Traditional reversed-phase sorbents (C8, C18) often struggle with complete phospholipid removal. Modern approaches favor mixed-mode sorbents that combine reversed-phase and ion-exchange mechanisms. Our MCX SPE cartridges exemplify this approach, offering both hydrophobic retention and strong cation exchange capabilities.
Specialized Phospholipid-Removal Sorbents
Advanced polymeric sorbents like HLB (Hydrophilic-Lipophilic Balance) have revolutionized phospholipid removal. Research shows that optimized HLB sorbents can remove over 95% of common matrix interferences including phospholipids, proteins, and salts (Waters Oasis Catalog, 2017). Our HLB SPE cartridges are specifically designed for this purpose, offering superior water-wettability and eliminating the need for conditioning steps.
pH Considerations for Sorbent Selection
The ionization state of both analytes and phospholipids significantly affects retention. For acidic analytes, maintaining a low pH (around 2.2) helps keep acidic drugs in their neutral forms while phospholipids remain charged. As demonstrated in comprehensive SPE procedures, “the starting pH of 2.2 results in less ionization of the acidic drugs and hence, better retention on the cartridge” (De Zeeuw & Franke, 2000).
Step 3: Wash Solvent Optimization
Systematic Wash Development
The wash step represents the critical juncture where phospholipids are removed while analytes remain retained. Begin optimization with increasing concentrations of methanol or acetonitrile in water, typically in 10% increments from 100% water to 100% methanol. As described in method development strategies, “for a simple non-polar extraction, the composition of the washing solvent should first be varied using increasing concentrations of methanol or acetonitrile in water” (Ingwersen, 2000).
pH-Modified Wash Solutions
For mixed-mode sorbents, incorporate pH adjustments in wash solvents. For basic analytes on MCX sorbents, acidic washes (2% formic acid in water) effectively remove neutral and acidic interferences while retaining basic compounds. The wash pH should remain at least 2 units away from the pKa of both analyte and sorbent to maintain ionic interactions.
Volume Optimization
Wash volume significantly impacts both phospholipid removal and analyte recovery. Studies show that excessive wash volumes can wash away polar acidic compounds. In comprehensive SPE procedures, researchers found that “the amounts of water in the sample application step and in the wash step should be kept as small as possible” to prevent loss of polar compounds (De Zeeuw & Franke, 2000).
Step 4: Evaluating LC-MS Matrix Effects
Post-Extraction Spike Experiments
The gold standard for evaluating matrix effects involves comparing analyte response in neat solution versus post-extraction spiked matrix. Calculate matrix factor (MF) using the formula: MF = (peak area in post-extracted spike) / (peak area in neat solution). Acceptable methods typically show MF values between 0.8-1.2, with CV < 15%.
Monitoring Phospholipid-Specific Ions
During method validation, continuously monitor characteristic phospholipid ions to ensure consistent removal. Modern LC-MS systems allow simultaneous monitoring of analyte and phospholipid ions, providing real-time feedback on extraction efficiency.
Ion Suppression Mapping
Create ion suppression maps by injecting blank extracts and monitoring suppression across the chromatographic run. Phospholipids typically elute in specific regions (often between 1-3 minutes in reversed-phase chromatography), and effective SPE methods should show minimal suppression in these regions.
Advanced Considerations for High-Throughput Applications
96-Well Plate Formats
For laboratories processing large sample batches, our 96-well SPE plates offer significant advantages. These formats maintain consistent phospholipid removal while improving throughput and reproducibility. As noted in SPE literature, “the 96 well plate format SPE devices have been adapted to remove precipitated proteins from biosamples” (Simpson, 2000), and this extends to phospholipid removal as well.
Automation Compatibility
Modern SPE methods must be compatible with automated liquid handling systems. Optimized wash protocols with consistent flow rates and volumes ensure reproducible phospholipid removal across automated platforms.
Practical Implementation Guidelines
Sample Preparation Protocol
- Plasma Pretreatment: Dilute plasma 1:1 with appropriate buffer (typically acidic for basic analytes, neutral to basic for acidic analytes)
- SPE Conditioning: For traditional sorbents, condition with methanol followed by water or buffer. For advanced sorbents like our HLB cartridges, conditioning may be eliminated
- Sample Loading: Apply at controlled flow rates (typically 1-2 mL/min)
- Wash Optimization: Use 2-3 mL of optimized wash solvent (e.g., 5% methanol in water for HLB, acidic wash for MCX)
- Elution: Use strong elution solvents (e.g., 90:10 acetonitrile:methanol for HLB, ammoniated methanol for MCX)
Troubleshooting Common Issues
If phospholipid removal remains inadequate:
- Increase wash strength: Incrementally increase organic content in wash solvent
- Add additional wash steps: Incorporate multiple wash solvents with different selectivities
- Optimize pH: Adjust sample and wash pH to maximize phospholipid removal while retaining analytes
- Consider sorbent alternatives: Switch to specialized phospholipid-removal sorbents like our WAX or WCX cartridges for challenging applications
Conclusion: Achieving Reliable Plasma Analysis
Effective phospholipid removal through optimized SPE represents a critical success factor in modern plasma analysis. By systematically identifying interference ions, selecting appropriate sorbent chemistry, optimizing wash conditions, and rigorously evaluating matrix effects, laboratories can achieve reliable, reproducible results. As the product manager at Poseidon Scientific, I recommend starting with our HLB SPE cartridges for general applications and our MCX SPE cartridges for basic compounds requiring enhanced phospholipid removal. For high-throughput laboratories, our 96-well SPE plates offer the perfect balance of performance and efficiency.
Remember that successful method development requires iterative optimization. Monitor both analyte recovery and phospholipid removal throughout the process, and don’t hesitate to contact our technical support team for assistance with challenging applications. With proper SPE methodology, phospholipid interference becomes a manageable challenge rather than an analytical roadblock.