Alkaloid Classes Commonly Found in Herbal Supplements
Herbal supplements contain diverse alkaloid classes that require specialized extraction methods for accurate analysis. The most common alkaloids found in these matrices include:
Quinolizidine Alkaloids
These compounds, such as those found in lupin seedlings, are well-retained by sulfonate SCX phases but may require mixed-mode approaches for optimal recovery. Research by Strobiecki et al. (1997) demonstrated successful separation using both SCX and C18 sorbents in tandem for complex plant matrices.
Benzophenanthridine Alkaloids
These compounds have been successfully extracted using C18 sorbents, as reported by Chauret et al. (1990). Their complex structures often benefit from mixed-mode retention mechanisms.
Catharanthus Alkaloids
Naaranlahti et al. (1990) documented effective extraction of these medicinally important alkaloids using C18 sorbents, highlighting the versatility of reversed-phase approaches for certain alkaloid classes.
Phenylethylamine Alkaloids
Compounds like hordenine, commonly found in green barley and various grasses, present extraction challenges due to their structural similarity to pharmaceutical compounds. These alkaloids are effectively retained by mixed-mode sorbents that combine ion-exchange and reversed-phase mechanisms.
It’s important to note that not all phytochemicals are equally amenable to SPE. Polyhydroxylated species such as aconine may show poor retention on mixed-mode sorbents due to inadequate initial retention by the reversed-phase mechanism. This variability underscores the importance of selecting appropriate sorbent chemistry for specific alkaloid classes.
Sample Extraction Using Acidified Methanol
The initial extraction step is critical for successful alkaloid isolation. Acidified methanol serves as an excellent extraction solvent for several reasons:
Solvent Selection Rationale
Methanol effectively penetrates plant cell walls and extracts a broad range of alkaloids. The acidification (typically with 0.1-1% formic acid or hydrochloric acid) serves multiple purposes:
- Protonates basic alkaloids, increasing their solubility in the extraction solvent
- Helps break down plant cell structures for more complete extraction
- Stabilizes alkaloids that might degrade under neutral or basic conditions
- Minimizes pH-catalyzed rearrangement or oxidation of labile compounds
Extraction Protocol
A typical extraction involves homogenizing 1-2 grams of herbal supplement material in 10-20 mL of acidified methanol (0.1% formic acid in methanol). The mixture should be vortexed vigorously for 1-2 minutes, then sonicated for 15-30 minutes at room temperature. Following sonication, the sample should be centrifuged at 3000-5000 × g for 10 minutes to separate particulate matter.
Research by Lenheer et al. (1984) demonstrated that lyophilized plant material ground and extracted with methanol, followed by filtration and evaporation, provides effective extraction of iridoid glucosides and flavanoids. This methodology can be adapted for alkaloid extraction with appropriate pH adjustments.
Filtration and Dilution Prior to SPE Loading
Proper sample preparation before SPE loading is essential for optimal cartridge performance and longevity.
Filtration Requirements
The methanolic extract should be filtered through a 0.45 μm or 0.22 μm syringe filter to remove particulate matter that could clog the SPE cartridge. For particularly complex matrices, additional filtration through glass microfiber filters may be necessary.
Dilution Strategy
The filtered extract typically requires dilution with water or aqueous buffer to achieve the optimal organic solvent concentration for SPE loading. For WCX cartridges, the final methanol concentration should be reduced to 10-20% (v/v) to ensure proper retention of protonated alkaloids.
The dilution also serves to adjust the sample pH to the optimal range of 5-6 for alkaloid retention on WCX sorbents. This pH adjustment is critical because WCX sorbents contain weak cation exchange groups (typically carboxylic acids) that effectively retain protonated bases when the pH is approximately 2 units below their pKa values.
Volume Considerations
The total sample volume loaded onto the SPE cartridge should not exceed the manufacturer’s recommendations. For a 60 mg WCX cartridge, typical loading volumes range from 1-5 mL, depending on the alkaloid concentration and matrix complexity.
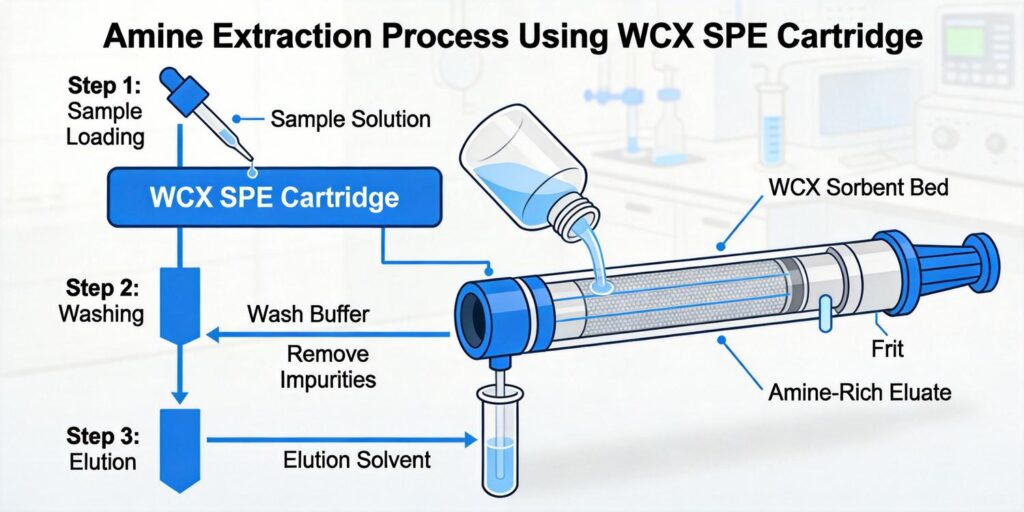
Conditioning WCX Cartridge with Methanol and Water
Proper conditioning of the WCX cartridge is essential for reproducible and efficient alkaloid recovery.
Conditioning Protocol
The standard conditioning sequence for WCX cartridges involves:
- Methanol Conditioning: Pass 2-3 mL of methanol through the cartridge at a flow rate of approximately 1 mL/min. This step solvates the polymeric backbone and prepares the sorbent for aqueous samples.
- Water Conditioning: Follow with 2-3 mL of deionized water or appropriate buffer at the same flow rate. This step removes methanol and creates an aqueous environment for sample loading.
Conditioning Importance
Proper conditioning ensures that the sorbent is fully activated and that the weak cation exchange groups are in their protonated form, ready to interact with basic alkaloids. Incomplete conditioning can lead to poor recovery and reproducibility issues.
Research by Bonazzi et al. (1995) demonstrated that for SCX sorbents (which share similar conditioning requirements with WCX), proper conditioning with methanol followed by buffer solution (pH 4.5) is essential for quantitative recovery of basic drugs like chlorhexidine.
Loading Extract at pH 5–6 to Retain Protonated Alkaloids
pH Optimization
The sample pH during loading is the most critical parameter for successful alkaloid retention on WCX sorbents. At pH 5-6:
- Most basic alkaloids (pKa > 8) are fully protonated and positively charged
- WCX carboxylic acid groups are partially ionized, creating effective cation exchange sites
- Interfering acidic and neutral compounds remain uncharged and pass through the cartridge
pH Adjustment Methods
The diluted extract should be adjusted to pH 5-6 using dilute acid (formic acid or acetic acid) or buffer solutions. Phosphate or acetate buffers at appropriate concentrations (10-50 mM) work well for maintaining consistent pH during loading.
Loading Flow Rate
Maintain a controlled flow rate of 1-2 mL/min during sample loading. Too fast a flow rate can reduce retention efficiency, while too slow can prolong the extraction process unnecessarily.
Studies have shown that basic drugs can be isolated from urine at neutral pH using mixed-mode cartridges, allowing extraction of compounds that might otherwise be unstable. This principle applies equally to alkaloid extraction from herbal matrices.
Washing with Methanol to Remove Pigments and Lipids
Wash Solvent Selection
Methanol serves as an effective wash solvent for several reasons:
- Removes non-polar interferences like pigments, lipids, and chlorophyll derivatives
- Does not disrupt the ion-exchange interaction between protonated alkaloids and WCX sorbent
- Evaporates easily during subsequent concentration steps
Wash Volume Optimization
Typically, 2-3 mL of methanol is sufficient to remove most interfering compounds. For particularly pigmented samples, additional wash volumes may be necessary. The wash step should continue until the eluent appears colorless or nearly so.
Alternative Wash Strategies
For samples with high lipid content, a sequential wash with methanol followed by methanol:water (90:10) can be more effective. Some protocols also incorporate a weak acid wash (1-2% formic acid in water) to remove weakly retained basic compounds before the methanol wash.
Historical approaches to plant material extraction often used SPE simply as a technique to remove pigments by passing lyophilized or homogenized samples through C18 devices. Modern WCX methods provide superior selectivity by combining reversed-phase and ion-exchange mechanisms.
Elution with Ammonia-Methanol Solution
Elution Mechanism
The elution step employs basic conditions to neutralize the positive charge on alkaloids, disrupting the ion-exchange interaction with the WCX sorbent. A typical elution solution contains 2-5% ammonium hydroxide in methanol.
Elution Protocol
- Apply 2-3 mL of ammonia-methanol solution to the conditioned and washed cartridge
- Allow the solution to remain in contact with the sorbent for 1-2 minutes to ensure complete alkaloid displacement
- Collect the eluate in a clean tube or vial
- Repeat with an additional 1-2 mL of elution solution to ensure complete recovery
Alternative Elution Solutions
While ammonia-methanol is most common, other basic eluents can be effective:
- 2% triethylamine in methanol
- Methanol containing 1% ammonium carbonate
- Basic methanol:acetonitrile mixtures (typically 90:10)
The choice of eluent may depend on the specific alkaloids being extracted and the subsequent analytical method.
Eluate Handling
The collected eluate should be evaporated to dryness under a gentle stream of nitrogen at 30-40°C. The residue can then be reconstituted in an appropriate solvent for LC-MS analysis, typically methanol or methanol:water mixtures compatible with the chromatographic system.
LC-MS Analysis of Alkaloid Composition
Instrumentation Considerations
Modern LC-MS systems provide the sensitivity and selectivity required for comprehensive alkaloid profiling:
- LC Conditions: Reverse-phase columns (C18 or equivalent) with gradient elution using water and methanol or acetonitrile, both containing 0.1% formic acid
- MS Detection: High-resolution mass spectrometry (HRMS) enables accurate mass measurements for compound identification
- Ionization: Electrospray ionization (ESI) in positive mode is most common for basic alkaloids
Method Validation
For quantitative applications, method validation should include:
- Linearity over the expected concentration range
- Limit of detection (LOD) and limit of quantification (LOQ)
- Recovery studies using spiked samples
- Precision (repeatability and reproducibility)
- Matrix effects evaluation
Data Analysis Strategies
Comprehensive alkaloid profiling often involves:
- Targeted analysis for known alkaloids using multiple reaction monitoring (MRM)
- Untargeted screening for novel or unexpected alkaloids using full-scan HRMS
- Metabolite profiling to identify alkaloid derivatives and transformation products
Quality Control Measures
Include appropriate quality controls in each analytical batch:
- Method blanks to monitor contamination
- Quality control samples at low, medium, and high concentrations
- Internal standards (preferably deuterated alkaloid analogs) to correct for matrix effects and instrument variability
WCX SPE Advantages for Alkaloid Isolation
Mixed-Mode Retention Mechanism
WCX sorbents offer distinct advantages for alkaloid isolation through their dual retention mechanism:
- Ion-exchange: Carboxylic acid groups retain protonated alkaloids through electrostatic interactions
- Reversed-phase: Polymeric backbone provides hydrophobic interactions for alkaloids with non-polar regions
This combination improves retention for all types of basic analytes, especially strong bases that might not be well-retained by traditional reversed-phase sorbents.
Selectivity Benefits
Compared to traditional solvent extraction or single-mode SPE, WCX provides:
- Superior removal of acidic and neutral matrix components
- Reduced co-extraction of pigments and lipids
- Cleaner extracts with lower matrix effects in LC-MS analysis
- Higher sensitivity through effective concentration of target alkaloids
Method Development Considerations
When developing WCX methods for specific herbal supplements:
- Determine the pKa range of target alkaloids to optimize loading pH
- Evaluate different wash solvents to maximize interference removal
- Test alternative elution solutions for complete alkaloid recovery
- Consider cartridge capacity relative to expected alkaloid concentrations
The Waters Oasis WCX material, for example, was specifically developed to provide better sample preparation for strong bases and quaternary amines, with a tightly controlled ion-exchange capacity ensuring reproducible SPE protocols.
Practical Considerations and Troubleshooting
Common Issues and Solutions
Poor Recovery
- Cause: Incorrect loading pH or insufficient conditioning
- Solution: Verify pH meter calibration and ensure proper conditioning sequence
Cartridge Clogging
- Cause: Inadequate sample filtration or excessive particulate matter
- Solution: Improve sample cleanup before SPE, consider larger particle size sorbents
Matrix Effects in LC-MS
- Cause: Incomplete removal of co-extractives
- Solution: Optimize wash steps, consider additional cleanup methods
Method Scalability
The WCX method described can be scaled for different sample sizes and throughput requirements:
- Small-scale: 10-30 mg cartridges for research applications
- Medium-scale: 60-150 mg cartridges for routine analysis
- High-throughput: 96-well plate formats for screening applications
Alternative SPE Formats
For laboratories with different requirements, consider:
- Poseidon Scientific WCX Cartridges: Available in various sizes and formats for flexible method development
- 96-well SPE Plates: Ideal for high-throughput applications and automation
- Mixed-mode alternatives: MCX for stronger cation exchange when dealing with weakly basic compounds
This comprehensive WCX SPE method provides a robust framework for alkaloid isolation from herbal supplements, offering superior selectivity and cleaner extracts compared to traditional extraction methods. The mixed-mode retention mechanism specifically addresses the challenges posed by basic alkaloids in complex plant matrices, making it an essential tool for quality control, research, and regulatory applications in the herbal supplement industry.