Understanding Solid Phase Extraction Fundamentals
Solid Phase Extraction (SPE) represents a cornerstone technique in modern analytical chemistry, offering a sophisticated yet accessible approach to sample preparation. As Dr. Xu, product manager at Poseidon Scientific, I’ve witnessed firsthand how mastering SPE fundamentals can transform analytical workflows across pharmaceutical, environmental, clinical, and food safety laboratories.
What Exactly Is Solid Phase Extraction?
At its core, SPE is defined as “bringing a liquid or gaseous test sample in contact with a solid phase, whereby the analyte is selectively adsorbed on the surface of the solid phase” (Simpson & Wells, 2000). This elegant process involves three fundamental elements: mass transport of molecules through a fluid to a surface, adsorption onto or partition into a solid phase, and selective desorption into a fluid with different properties.
Unlike traditional liquid-liquid extraction, SPE offers several distinct advantages: improved throughput through parallel processing, decreased organic solvent usage, higher and more reproducible recoveries, cleaner extracts without emulsion formation, tunable selectivity through phase choices, and ready automation capabilities.
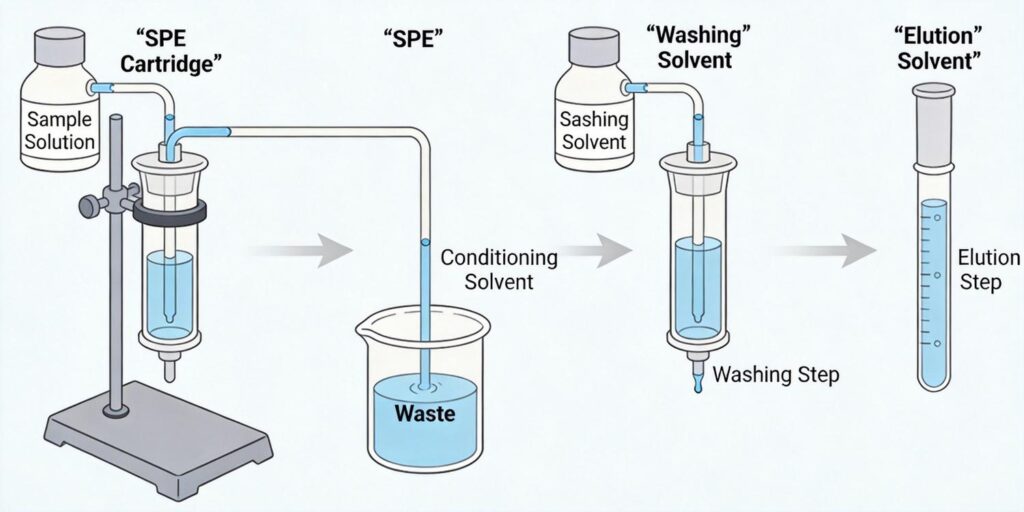
The Core SPE Process: Five Essential Steps
Most SPE procedures follow five fundamental steps that form the backbone of successful extractions:
1. Conditioning: Preparing the Sorbent Surface
Conditioning serves multiple critical functions. First, it solvates the SPE column and normalizes the column environment to the sample, while removing microparticulates and interferents. For hydrophobic phases like our C18 SPE cartridges, methanol followed by water is typically used. This process expands functional binding sites away from the solid surface, exposing them to diffusive flow. Proper conditioning ensures optimal contact and binding of the sample, with flow rates between 0.5-3.0 mL/min generally acceptable for sufficient solvent-sorbent contact without causing channeling.
2. Sample Application: Loading Your Analyte
During sample application, the liquid sample containing analytes of interest is introduced to the sorbent. Analytes interact with the sorbent material and are retained, while undesired matrix components ideally pass through. This step involves diffusion of sample through the sorbent bed for binding of target compounds to sorbent functional groups. Driving forces for sample loading include gravity, pressure, and vacuum, with optimal application rates typically around 1 mL/min for maximum recovery.
3. Washing: Removing Interfering Compounds
Washing steps remove excess sample matrix and unretained compounds that may have been retained along with the analytes of interest. The washing solvent is carefully chosen to elute impurities while leaving target analytes bound to the sorbent. This step involves passing a solvent that won’t elute the analyte but will remove weakly bound matrix components.
4. Drying: Preparing for Elution
This optional but often crucial step removes aqueous or immiscible solvents to prepare the sorbent for the elution solvent. Drying is particularly important when elution solvents are immiscible with wash solvents and sample matrices.
5. Elution: Recovering Your Analyte
Finally, a suitable solvent (or series of solvents) disrupts functional binding mechanisms and selectively releases compounds for collection and analysis. This step results in a purified and often concentrated extract containing the compounds of interest. The elution solvent provides a more desirable environment for the analyte than the solid phase, causing desorption and collection in the liquid as it exits the SPE device.
SPE Bonding Mechanisms and Sorbent Chemistry
Understanding SPE mechanisms is essential for method development. Based on sorbent material, there are three main types of SPE mechanisms:
Reversed-Phase SPE (Hydrophobic Bonding)
Reversed-phase SPE, also called nonpolar bonding, occurs via attraction of like hydrocarbon groups through van der Waals forces or dispersion forces. Bond strength is relatively weak at 1–5 kcal/mol, with increasing strength as hydrocarbon atoms increase. Hydrophobic phases include alkyl chains from C2 to C18, with capacity dependent on alkyl chain configuration and total carbon loading on the silica substrate. Our HLB SPE cartridges offer excellent reversed-phase performance for a wide range of applications.
Normal Phase SPE (Polar Bonding)
Normal phase SPE utilizes polar interactions such as hydrogen bonding, dipole-dipole interactions, and π-π interactions. These sorbents are effective for extracting polar compounds from nonpolar matrices.
Ion-Exchange SPE
Ion-exchange SPE involves electrostatic interactions between charged analytes and oppositely charged sorbent functional groups. This includes strong cation exchange (SCX) and strong anion exchange (SAX) mechanisms. Our MCX SPE cartridges and WAX SPE cartridges provide excellent ion-exchange capabilities for charged analytes.
Mixed-Mode SPE
Mixed-mode sorbents combine multiple retention mechanisms (typically reversed-phase and ion-exchange) for enhanced selectivity. These are particularly valuable for complex matrices where multiple interaction types are needed for optimal cleanup.
Key Sorbent Materials and Their Properties
Successful SPE requires understanding sorbent characteristics. Desirable properties of SPE particles include:
- Porous structure with large surface area (>100 m²/g)
- Reversible adsorption capabilities
- High purity with low impurities
- Good chemical stability
- Excellent surface contact with sample solutions
- High percentage recovery
Common sorbent materials include bonded silica phases, organic polymers, modified polymeric sorbents with specific functional groups, porous graphitized carbons, and ion exchange sorbents. Silica-based sorbents dominate the market due to their availability in well-defined surface areas and pore sizes, relatively low cost, and versatile surface chemistry dominated by silanol groups.
Method Development and Optimization Basics
Developing an effective SPE method requires systematic optimization. A general SPE method development strategy includes:
Background Research and Characterization
Begin by researching previous SPE and analysis conditions for your analyte and matrix. Characterize the analyte’s structure, pKa, polarity, functional groups, solvent solubility, and stability. Simultaneously, characterize the sample matrix for possible interferences, pH, ionic strength, and qualitative/quantitative variability.
Experimental Optimization
Develop or apply effective HPLC or GC conditions to monitor progress, assessing recovery and eluate cleanliness. Select and test sorbents to determine which provide maximum analyte retention and which eluent solvents yield highest recoveries. Identify optimum wash solvents that assess eluate cleanliness under conditions of maximum analyte retention.
Method Validation Considerations
Validate your SPE procedure by testing variables including sorbent weight, different cartridges and lots, preconditioning solvents, loading solvent composition (% organic, pH, ionic strength, volume), wash solvent parameters, eluent volume and composition, flow rates during loading, wash, and elution, linearity and range, and analyte/matrix stability.
Practical Considerations for New Analytical Chemists
For those new to SPE, several practical considerations can significantly impact success:
Flow Rate Management
Maintain appropriate flow rates throughout the SPE process. Excessive flow can cause channeling in the sorbent bed, reducing available surface area and recovery. Flow rates between 0.5-3.0 mL/min are generally acceptable for most applications.
pH and Ionic Strength Optimization
For reversed-phase SPE, un-ionized compounds favor retention. Ensure the pH of the sample and sorbent are equivalent for optimal binding. For ion-exchange SPE, analytes must carry the opposite charge of the sorbent functional groups.
Sample Preparation
Remove particulates if necessary (by centrifugation or filtering) and dilute viscous matrices with water or buffer to ensure proper pH for desired interactions. Always wash the column with the strongest solvent in which it will be placed during extraction to minimize background noise.
Common SPE Formats and Applications
SPE is available in various formats to suit different laboratory needs:
- Cartridges: The most common format, available in sizes from 100-1000 mg with 1-6 mL tube capacities
- 96-Well Plates: Ideal for high-throughput applications and automation
- Disks: Recommended for large volume samples, samples containing high amounts of particulates, or when high flow rates are required
SPE finds applications across numerous industries, including pharmaceutical compounds and metabolites in biological fluids, drugs of abuse testing, environmental pollutants in water, pesticides and antibiotics in food matrices, desalting proteins and peptides, lipid fractionation, and vitamin analysis.
Troubleshooting Common SPE Issues
Even with proper technique, challenges can arise. Common issues include:
Breakthrough
Breakthrough occurs when analytes pass through the column unretained due to weak attachment or exceeded column capacity. Causes include improper conditioning, excessive flow rates, incorrect pH, or insufficient sorbent capacity.
Low Recovery
Low recovery can result from incomplete elution, analyte degradation, or irreversible binding to the sorbent. Optimize elution solvent strength and volume, and consider adding modifiers to improve recovery.
Poor Cleanup
Insufficient cleanup often stems from inadequate washing steps or incorrect wash solvent selection. Optimize wash conditions to remove interferences while retaining target analytes.
Conclusion: Building Your SPE Expertise
Solid Phase Extraction represents a powerful, versatile technique that, when mastered, can significantly enhance analytical capabilities. By understanding the fundamental principles, sorbent chemistries, and optimization strategies outlined here, new analytical chemists can develop robust, reproducible SPE methods tailored to their specific applications.
At Poseidon Scientific, we offer a comprehensive range of SPE products including HLB, MAX, MCX, WAX, WCX, and 96-well SPE plates to support your analytical needs. Remember that successful SPE requires balancing sensitivity (recovery) with selectivity (cleanliness), with the ultimate goal of achieving acceptable limits of detection with minimal interfering compounds.
As you develop your SPE expertise, focus on method performance based on optimal recovery needed to sustain required signal-to-noise ratios, not just absolute percent recovery. With practice and attention to detail, SPE will become an indispensable tool in your analytical chemistry toolkit.