Common Forensic Drugs Analyzed with MCX SPE
Mixed-mode cation exchange (MCX) solid phase extraction has become a gold standard in forensic toxicology laboratories for analyzing basic drugs and their metabolites. The most commonly targeted compounds include amphetamine and methamphetamine (phenylethylamine derivatives) and cocaine metabolites such as benzoylecgonine and norcocaine. These substances are potent central nervous system stimulants that require precise analytical methods for accurate detection and quantification in biological matrices.
According to established forensic protocols, amphetamine and methamphetamine analysis typically targets quantification ions at m/z 240 and 254 respectively, while cocaine metabolites are monitored at specific ion ratios for confirmation. The metabolism of these compounds follows distinct pathways—methamphetamine metabolizes to amphetamine, while cocaine undergoes hydrolysis to benzoylecgonine and other metabolites that require specialized extraction approaches.
Sample Matrices: Blood and Urine Considerations
Forensic drug analysis primarily focuses on two biological matrices: urine and whole blood. Each presents unique challenges for MCX SPE extraction:
Urine Samples
Urine represents the most common forensic matrix due to its non-invasive collection and relatively high drug metabolite concentrations. Standard protocols typically use 2-5 mL urine aliquots, with pH adjustment to 6.0 ± 0.5 using phosphate buffer. The presence of glucuronide conjugates often necessitates hydrolysis steps before extraction.
Whole Blood Samples
Blood analysis requires more extensive pretreatment due to protein binding and cellular components. Standard procedures involve diluting 2 mL of blood with 8 mL of deionized water, followed by centrifugation to remove cellular debris. The resulting supernatant is then pH-adjusted to between 4.8 and 5.5 using acetic acid before MCX SPE extraction.
Sample Hydrolysis and Pretreatment Protocols
Many forensic drugs, particularly opiates and benzodiazepines, exist as glucuronide conjugates in biological matrices. Effective hydrolysis is essential for accurate quantification:
Acid Hydrolysis for Opiates
For opiate analysis, standard protocols involve adding 400 μL of concentrated HCl and 200 μL of 10% hydroxylamine solution to 2 mL urine, followed by heating at 90°C for 20 minutes or autoclaving for 15 minutes on a liquid cycle. This process cleaves glucuronide bonds while stabilizing certain metabolites through oxime formation.
Enzymatic Hydrolysis
Alternative approaches use β-glucuronidase enzymes for gentler hydrolysis, particularly for heat-sensitive compounds. The choice between acid and enzymatic hydrolysis depends on the target analytes and required sensitivity levels.
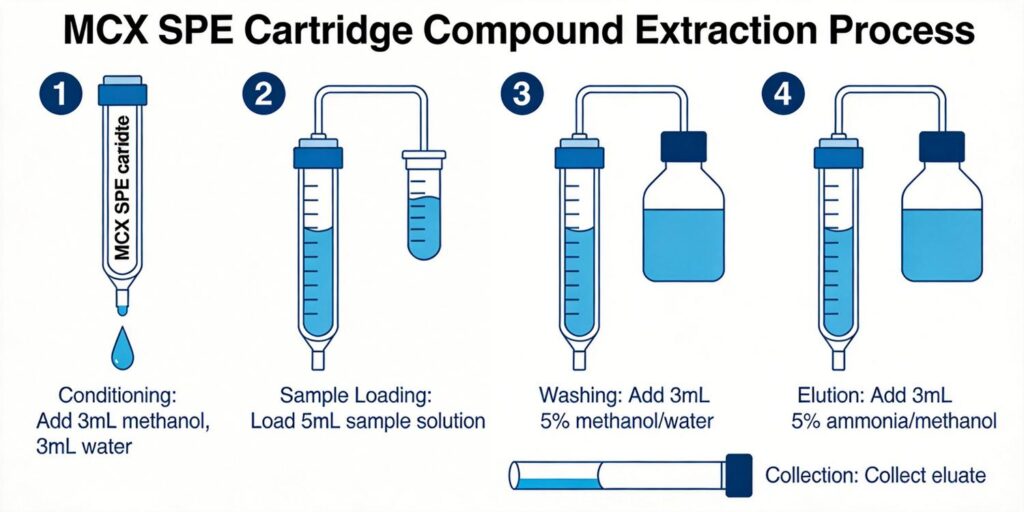
MCX Cartridge Conditioning Protocol
Proper conditioning of MCX cartridges is critical for optimal recovery and reproducibility. Standard conditioning protocols follow this sequence:
- 3 mL methanol to activate the hydrophobic phase and remove any residual contaminants
- 3 mL deionized water to transition the sorbent to aqueous conditions
- 1 mL of 0.1 M phosphate buffer, pH 6.0 or 0.1 M acetic acid to prepare the cation exchange sites
Critical note: Aspiration should be performed at ≤3 in. Hg to prevent sorbent drying, which can compromise extraction efficiency. For automated systems like RapidTrace, specific flow rates (typically 18 mL/min for conditioning steps) ensure consistent performance.
Loading Acidic Samples for Protonated Analytes
The MCX mechanism relies on maintaining analytes in their protonated (positively charged) forms during sample loading. For basic drugs like amphetamines and cocaine metabolites:
pH Optimization
Sample pH should be adjusted to 4.8-5.5 using acetic acid or phosphate buffer. At this pH range, basic drugs (pKa typically 8-10) remain protonated and interact strongly with the sulfonic acid cation exchange sites on the MCX sorbent.
Loading Parameters
Optimal loading occurs at 1-2 mL/min flow rates. For urine samples, typical loading volumes are 3-5 mL after pH adjustment and buffer addition. For blood samples, the diluted supernatant (approximately 10 mL) is loaded following the same pH principles.
Multi-Step Wash to Remove Endogenous Compounds
Effective washing removes interfering endogenous compounds while retaining target analytes. Standard MCX wash protocols include:
- 3 mL deionized water to remove water-soluble salts and polar interferences
- 1-2 mL of 0.1 M acetic acid or 0.1 M HCl to maintain acidic conditions and remove weakly bound compounds
- 3 mL methanol to eliminate hydrophobic interferences through the reversed-phase mechanism
Following the methanol wash, columns are typically dried under vacuum (5 minutes at ≥10 in. Hg) to remove residual methanol before elution. Some protocols include additional washes with hexane or acetonitrile/water mixtures for specific applications.
Basic Organic Elution Solvent Formulation
Elution disrupts the ionic interactions between protonated analytes and cation exchange sites through pH manipulation and organic solvent strength:
Standard Elution Solvent
The most common elution formulation for basic drugs is methylene chloride-isopropanol-ammonium hydroxide (78:20:2 v/v/v). This combination provides:
- Methylene chloride: Strong elution strength for hydrophobic interactions
- Isopropanol: Modifier to improve wettability and elution efficiency
- Ammonium hydroxide: Raises pH to 11.0-12.0, deprotonating analytes and disrupting ionic bonds
Alternative Elution Options
Some protocols use methanol-ammonium hydroxide (98:2) followed by liquid-liquid extraction with methylene chloride. This approach may be preferred for certain instrumentation compatibility requirements.
Critical note: Elution solvents should be prepared fresh daily, with isopropanol added to ammonium hydroxide first, followed by methylene chloride, to ensure proper pH (≥11.0).
GC-MS vs LC-MS Compatibility Considerations
The choice between gas chromatography-mass spectrometry (GC-MS) and liquid chromatography-mass spectrometry (LC-MS) influences MCX SPE protocol optimization:
GC-MS Compatibility
For GC-MS analysis, eluates typically require:
- Evaporation to dryness at ≤40°C under nitrogen
- Derivatization with reagents like BSTFA (with 1% TMCS) or MBHFBA
- Reaction at 70°C for 10-20 minutes depending on analytes
- Specific ion monitoring (e.g., m/z 240 for amphetamine, m/z 371 for carboxy-THC)
LC-MS Compatibility
For LC-MS applications:
- Eluates may be evaporated and reconstituted in mobile phase
- No derivatization typically required
- Methanol-based elution may be preferred over chlorinated solvents
- pH adjustment before injection may be necessary
Instrument-Specific Considerations
GC-MS methods often use capillary columns (30 m × 0.25 mm × 0.25 μm) with temperature programming from 60°C to 300°C at 25°C/min. LC-MS methods require consideration of ionization compatibility—basic eluents may suppress electrospray ionization for certain compounds.
Practical Implementation Notes
When implementing MCX SPE for forensic drug analysis, consider these key factors:
- Internal standards: Deuterated analogs (e.g., D5-amphetamine, D5-methamphetamine) provide optimal quantification accuracy
- Quality controls: Include at least three concentration levels covering the analytical range
- Matrix effects: Evaluate ion suppression/enhancement for LC-MS methods
- Carryover prevention: Implement adequate washing between samples in automated systems
For laboratories considering MCX SPE implementation, Poseidon Scientific’s MCX cartridges offer reliable performance with consistent lot-to-lot reproducibility. The mixed-mode mechanism provides superior selectivity compared to single-mode sorbents, particularly for complex forensic matrices.
Related extraction approaches include WAX (weak anion exchange) for acidic compounds and HLB (hydrophilic-lipophilic balance) for broader spectrum applications. For high-throughput laboratories, 96-well SPE plates provide automation compatibility while maintaining extraction efficiency.